| OCR Text |
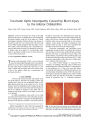
Show ORIGINAL CONTRIBUTION Topical Apraclonidine Testing Discloses Pupillary Sympathetic Denervation in Diabetic Patients Feray Koc, MD, Tulay Kansu, MD, Sevim Kavuncu, MD, and Esin Firat, MD Background: Autonomic denervation is common in diabetes mellitus ( DM). Pupillary sympathetic denervation ( PSD) has been found in Horner syndrome following instillation of apraclonidine 0.5%. We have applied this technique to investigate the prevalence of PSD in DM. Methods: Apraclonidine 0.5% was instilled in the eyes of 50 patients with DM and 30 age- matched and gender-matched subjects without DM ( control subjects). Pupil diameters ( PD) were measured before and 60 minutes after instillation. The duration of DM and the degree of diabetic retinopathy ( DR) were recorded for each patient. Results: Apraclonidine instillation caused an average of 0.9 mm of mydriasis ( range 0 to 4.5 mm) in DM and - 0.1 mm miosis ( range 0.5 to - 1 mm) in control subjects ( P < 0.001). Mydriasis of at least 1 mm was observed in 42% of DM patients. The change in PD was highly correlated with the duration of DM ( r = 0.368, P = 0.008) and the presence of DR ( r = 0.532, P < 0.001). Conclusion: Apraclonidine testing, which is easy to perform and not distressing to the patient, identified PSD in nearly half of DM patients, the degree of mydriasis being correlated to the duration of DM and the presence of DR. ( J Neuro- Ophthalmol 2006; 26: 25- 29) Autonomic neuropathy in diabetes mellitus ( DM) often develops insidiously. Its symptoms are vague and its signs are difficult to detect on routine physical examination. The presence of cardiac autonomic neuropathy has been found to increase mortality significantly ( 1- 3). In the early stages, autonomic and somatic peripheral neuropathy are reversible through improved glycemic control ( 4,5). The Diabetes Control and Complications Trial data clearly Neuro- Ophthalmology Unit ( FK, KS, EF), SB Ulucanlar Eye Hospital, Department of Neurology, Neuro- Ophthalmology Unit ( TK), 2Hacettepe University Faculty of Medicine, Ankara, Turkey. This study was conducted in Hacettepe University Hospitals and SB Ulucanlar Eye Hospital, under the approval of our institutional ethic committee. There is no competing interest. Address correspondence to Feray Koc, MD, Kuzgun sok 48/ 3 Asagi Ayranci Ankara 06540 Turkey; E- mail: dr_ feray@ yahoo. com support the benefit of intensive therapy in preventing their appearance ( 6). Thus, screening for autonomic dysfunction would be valuable. The pupil is a good site to obtain information about the state of diabetic autonomic neuropathy because the innervation of the pupil is exclusively autonomic and it is easily accessible for study without causing any discomfort to the patient. Infrared television pupiUography is a useful device to evaluate pupillary function but expensive and not widely available ( 3). A more practical method is to use topical pharmacologic agents. Previous studies of topical pharmacologic testing for autonomic pupillary dysfunction have shown inconsistent results ( 7- 11). Apraclonidine has been found to dilate the pupil in Horner syndrome but not more than 0.5 mm in normal eyes ( 12,13). In an earlier study ( 14), we found that apraclonidine 0.5% had a sensitivity and specificity equivalent to topical cocaine in the diagnosis of Horner syndrome. It caused at least 1 mm of pupil dilation 60 minutes after instillation in eyes with cocaine- confirmed Horner syndrome, but it did not cause more than 0.5 mm dilation in any control eyes. In this study, we used apraclonidine 0.5% to screen for pupillary sympathetic denervation ( PSD) in DM and tested the strength of the correlation between PSD, the presence of diabetic retinopathy ( DR), and the duration of DM. METHODS Patients with DM were drawn from those seeking a routine ophthalmologic examination in the ophthalmology clinic between March and June 2005. Subjects were excluded if they had an ocular pathologic condition apart from diabetic retinopathy, a contraindication to the use of ^- adrenoceptor antagonists or a- adrenoceptor agonists, previous intraocular surgery, use of contact lenses within three days before starting the study, or use of a systemic adrenergic medication within 15 days of test administration. Fifty patients with DM were recruited. Thirty healthy volunteers without DM who otherwise had the same inclusion criteria made up the control group. The evaluation of mydriasis was restricted to the right eyes in J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 25 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 Koc et al each group. The duration of the DM and the degree of metabolic control were recorded for each patient. Standard ophthalmologic examinations were performed first. Baseline pupil diameters ( PD) were determined to the nearest 0.5 mm using the pupil gauge on the Rosenbaum pocket vision screener in normal room lighting. The same measurements were repeated 60 minutes after instillation of one drop of apraclonidine 0.5%. After completing the apraclonidine test, fundus examinations were done under the slit- lamp and the retinal findings were recorded as diabetic retinopathy ( DR) absent, background diabetic retinopathy ( BDR) present, or proliferative diabetic retinopathy ( PDR) present. PD before and 60 minutes after apraclonidine instillation and the change in PD were compared in the two groups. The Independent samples t test, the x2 test, and the Pearson Correlation analysis were used in the statistical analysis. The study was approved by our institutional ethics committee and consent was taken from each patient before test administration. RESULTS The DM and control groups were not different with respect to age and gender ( Table 1). Twenty- two patients were insulin- dependent, 26 were using oral hypoglycemic agents, and two were using dietary control alone. Fundus examination showed that 28 patients had no DR, 10 had BDR, and 12 had PDR. The initial mean PD was significantly smaller in the DM patients than in the control subjects ( P < 0.001). After instillation of apraclonidine, this situation was reversed. The average mydriasis in DM patients was 0.9 mm ( range 0- 4.5 mm). In the DM patients, 30 patients developed mydriasis of 0.5- 4.5 mm, 18 had no change in PD, and two developed miosis. Twenty- one ( 42%) patients in the DM group showed a mydriatic response of s 1 mm which is diagnostic for PSD ( 14) ( Figs. 1, 2). In the control group, apraclonidine instillation caused miosis in 12 subjects, did not change the PD in 13 subjects, and caused slight mydriasis ( up to 0.5 mm) in 5 subjects. The average change in PD among control subjects was 0.1 mm ( range 0.5 mm mydriasis to 1 mm miosis) of miosis ( Fig. 2). In the diabetic patients, the mydriatic effect was correlated with the duration of DM ( r = 0.368, P = 0.008) ( Fig. 2) and with the presence of DR ( r = 0.532, P < 0.001) ( Fig. 3). The majority of the patients showing PSD were insulin- dependent ( 16/ 21) and had coexistent DR ( 17/ 21). The patients displayed no adverse effects from the apraclonidine test except 1- 4 mm upper lid retraction which developed in 64% of the DM patients and in 40% of the control subjects. DISCUSSION In the present study, we found that apraclonidine caused at least 1 mm of mydriasis in 42% of eyes with DM and in no eyes of control subjects. The degree of mydriasis in DM was correlated with the duration of diabetes and the presence of retinopathy. This finding suggests a high prevalence of PSD in DM, especially when it has been present a long time and has caused retinopathy. Apraclonidine modulates the effect of endogenous neurotransmitters through pre- junctional and postjunctional a2- receptors and would be expected to cause pupillary miosis in normal subjects ( 15,16). But it also has the ability to stimulate a x- adrenergic receptors with a lower efficacy than norepinephrine ( 12). In a study to determine its site of action in lowering intraocular pressure, apraclonidine was found to cause mydriasis in patients who had both glaucoma and Horner syndrome ( 12). This effect was explained by denervation hypersensitivity to apraclonidine of arreceptors in the iris dilator muscles because of loss of normal sympathetic innervation ( 12,17). Reversal of anisocoria in apraclonidine testing is now taken as a criterion for the diagnosis of Horner syndrome. However, in diabetic autonomic neuropathy, pupils are often affected bilaterally so it is not possible to use a change in anisocoria or to use the untreated eye as a control in TABLE 1. Demographic and pupillary data of the diabetes mellitus patients and control subjects Diabetes mellitus ( N = 50) Control subjects ( N = 30) P value Age ( years) Gender ( F/ M) Baseline pupil diameter ( mm) Pupil diameter ( mm) 60 minutes after apraclonidine 0.5% Change in pupil diameter ( mm) after apraclonidine 52.2 ± 13.0 29/ 21 2.5 ± 0.5 3.5 ± 1.2 0.9 ± 1.2 49.9 ± 11.1 14/ 16 3.0 ± 0.6 2.9 ± 0.4 - 0.1 ± 0.4 0.425* 0.325f < 0.001* 0.01* < 0.001* * Independent samples t test; f x 26 © 2006 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. Apraclonidine in Diabetes J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 FIG. 1. Bilateral pupillary sympathetic denervation in a diabetic patient demonstrated with topical apraclonidine 0.5%. A. Baseline condition shows bilateral miosis. B. There is no change in pupil size 60 minutes after instillation of cocaine 10%. C. There is bilateral mydriasis 60 minutes after instillation of apraclonidine 0.5%. E ^ E^ L_ CO CD E = 2- Q- : 3 Q. £ 1 CD CD CO 0 c o I 1 N = 25 ID 12 None Background Proliferative Diabetic Retinopathy FIG. 3. Change in pupil diameter following instillation of apraclonidine 0.5% in the eyes of diabetic patients according to presence of retinopathy. ( Each box shows the median, quartiles, and extreme values within a category; N, Number of patients in each category.) doing pharmacological testing. To detect PSD, one must know the effects of the testing agent on normal pupils. In this study, we observed no change in the pupil diameter in the majority of control eyes. Although we did not evaluate our patients critically in this respect, diabetic patients could have many ocular surface problems like keratoepitheliopathy, decreased corneal sensation, decreased tear film break- up time, and Duration of diabetes mellitus ( years) FIG. 2. Change in pupil diameter following instillation of apraclonidine 0.5% in the eyes of control subjects and diabetic patients according to duration of diabetes. ( Each box shows the median, quartiles, and extreme values within a category; N, Number of patients in each category.) decreased reflex tear secretion ( 18- 20). These changes may result in higher penetration of apraclonidine to the pupillary area in diabetic eyes as compared to healthy eyes through an impaired corneal barrier mechanism or through slow clearance from the tear film ( 21). The potential difference in bioavailability of apraclonidine in diabetic and healthy eyes might confound our results. However, in previous studies ( 12,13,22,23) the effect of topical apraclonidine on pupil size was found to be dose- independent at 0.25% to 1% and our apraclonidine concentration ( 0.5%) was within this range. By contrast, the intraocular pressure- lowering effect of apraclonidine appears to be dose- dependent ( 22- 25). Although we did not apply it in our study, monitoring of intraocular pressures during testing would have been useful. Had apraclonidine lowered intraocular pressure more in diabetics, corneal permeability might have played a role. Previous studies have shown that the response to sympathomimetic amines in healthy subjects is dependent on their age and there are no reported absolute values described for the investigated pharmacological pupil function tests ( 9,10). Smith and Smith ( 10) and Clark ( 9) have studied pupil denervation hypersensitivity in diabetic patients using different concentrations of phenylephrine, but these studies lacked an accurate assessment of the normal pupillary response within age groups. In those studies, the mydriatic effect of phenylephrine was highly variable and significantly correlated with the age of the patient ( r = 0.76, P < 0.001 [ 10]; r = 0.64, P < 0.001 [ 9]). A similar correlation between the mydriatic effect of hydroxyamphetamine and age has also been found, but the 27 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 Koc et al TABLE 2. Results of reported studies of topical pharmacologic testing for pupillary sympathetic denervation in diabetes Study Subjects Duration of diabetes ( average in years) Topical agent Results Smith et al, 1983 ( 10) Clark, 1988 ( 9) Cahill et al, 2001 ( 8) Pittasch et al, 2002 ( 11) Current series 34 28 141 31 50 15.9 20.9 Not reported 14.2 10.6 Phenylephrine 2% Hydroxyamphetamine 0.5% Phenylephrine 0.5% Cocaine 4% Cocaine 4% Epinephrine 1% Pholedrine 5% Apraclonidine 0.5% 47% PSD Normal pupillary dilation 38% PSD Normal pupillary dilation Reduced pupillary dilation Normal pupillary dilation Reduced pupillary dilation 42% PSD PSD, pupillary sympathetic denervation. influence of age is less marked ( r = 0.32, P < 0.05 [ 10]). An age- dependent reduction in cocaine response has been observed in previous studies ( 8,26). We also observed age dependency in the apraclonidine response in our study, but the strength of the correlation was less significant ( r - 0.22, P = 0.05). Less age dependency allows us to define the limits of normal and pathologic responses more accurately, and this is an advantage of apraclonidine in screening of PSD. Previous reports concerning pupillary sympathetic function as demonstrated with different pharmacological agents in diabetic patients are given in Table 2 ( 8- 11). In all studies, the effect of the testing agent was compared between diabetics and age- matched controls. Smith and Smith ( 10) and Clark ( 9) reported their results as percentages of patients showing PSD. Smith and Smith ( 10) used phenylephrine 2% and found a prevalence of PSD equivalent to our results. Clark ( 9) used phenylephrine 0.5% and found a lower prevalence of PSD even though patients had a longer duration of diabetes and all had PDR. The concentration of the testing agent ( 0.5%) might explain the relatively low prevalence. Pittasch ( 11) demonstrated a significant reduction in the normal mydriatic response to cocaine 4% and pholedrine 5% in diabetic groups, but he could not demonstrate significant denervation hypersensitivity in those groups to epinephrine 1%. With its selectivity for a i - adrenergic receptors, phenylephrine is an effective direct adrenergic agonist for dilating the pupil by stimulating the dilator muscle. Because it stimulates both a r and a2- adrenergic receptors, which have counteracting effects, epinephrine is a less potent mydriatic agent. Perhaps epinephrine 1% is not sensitive enough to detect denervation hypersensitivity. In performing cocaine testing, Cahill ( 8) could not demonstrate significant PSD in diabetics, but they had relatively mild diabetic changes. Previous studies have established a direct correlation between the degree of cardiovascular autonomic neuropathy and the severity of retinopathy ( 27,28). We observed a similar correlation between PSD and the severity of retinopathy, the prevalence of PSD being 14%, 70%, and 83% in patients without DR, with BDR, and with PDR, respectively. Because we did not select our study group randomly, our results are not an accurate measure of the true prevalence of PSD in diabetes, but they do confirm that autonomic neuropathy is very common in long- term diabetics. Considering the increased risk of mortality with diabetic autonomic neuropathy, screening for it with a simple noninvasive method is valuable. REFERENCES 1. McNally PG, Lawrence IG, Panerai RB, et al. Sudden death in type- 1 diabetes. Diabetes Obes Metab 1999; 1: 151- 8. 2. Ewing DJ, Campbell IW, Clarke BE The natural history of diabetic autonomic neuropathy. J Med 1980; 49: 95- 108. 3. Ewing DJ, Clarke BE Autonomic neuropathy: Its diagnosis and prognosis. Clin Endocrinol Metab 1986; 15: 855- 88. 4. Hreidarsson AB. The pupil of the eye in diabetes mellitus, an indicator of autonomic nervous dysfunction. Dan Med Bull 1992; 39: 400- 8. 5. Terkildsen AB, Christensen NJ. Reversible nervous abnormalities in juvenile diabetics with recently diagnosed diabetes. Diabetologia 1971; 7: 113- 7. 6. Santiago TV Lessons from the Diabetes Control and Complications Trial. Diabetes 1993; 42: 1549- 54. 7. Sharma S, Hoskin- Mott A, Benstead T, et al. Correlation of the pilo-pupil ratio average, a new test for autonomic denervation, to the severity of diabetic retinopathy. Can J Ophthalmol 1997; 32: 170^ 4. 8. Cahill M, Eustace P, De Jesus V Pupillary autonomic denervation with increasing duration of diabetes mellitus. Br J Ophthalmol 2001; 85: 1225- 30. 9. Clark CV Ocular autonomic nerve function in proliferative diabetic retinopathy. Eye 1988; 2: 96- 101. 10. Smith SA, Smith SE. Evidence for a neuropathic aetiology in the small pupil of diabetes mellitus. Br J Ophthalmol 1983; 67: 89- 93. 11. Pittasch D, Lobmann R, Behrens- Baumann W, et al. Pupil signs of sympathetic autonomic neuropathy in patients with type 1 diabetes. Diabetes Care 2002; 25: 1545- 50. 12. Morales J, Brown SM, Abdul- Rahim AS, et al. Ocular effects of apraclonidine in Horner syndrome. Arch Ophthalmol 2000; 118: 951- 4. 28 © 2006 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. Apraclonidine in Diabetes J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 13. Brown SM, Aouchiche R, Freedman KA. The utility of 0.5% apraclonidine in the diagnosis of Horner syndrome. Arch Ophthalmol 2003; 121: 1201- 3. 14. Koc F, Kavuncu S, Kansu T, et al. The sensitivity and specificity of 0.5% apraclonidine in the diagnosis of oculosympathetic paresis. Br J Ophthalmol 2005; 89: 1442^ 1. 15. Crosson CE, Heath AR, DeVries GW, et al. Pharmacological evidence for heterogeneity of ocular alpha 2 adrenoceptors. Curr Eye Res 1992; 11: 963- 70. 16. Potter DE, Crosson CE Heath AR, et al. Review: alpha 2 DA2 agonists as antiglaucoma agents: comparative pharmacology and clinical potential. J Ocul Pharmacol 1990; 6: 251- 7. 17. Langham ME, Weinstein GW. Horner's syndrome: ocular supersensitivity to adrenergic amines. Arch Ophthalmol 1967; 78: 462- 9. 18. Yoon KC, Im SK, Seo MS. Changes of tear film and ocular surface in diabetes mellitus. Korean J Ophthalmol 2004; 18: 168- 74. 19. Saito J, Enoki M, Hara M, Morishige N, Chikama T, Nishida T Correlation of corneal sensation, but not of basal or reflex tear secretion with the stage of diabetic retinopathy. Cornea 2003; 22: 15- 8. 20. Inoue K, Kato S, Ohara C, et al. Ocular and systemic factors relevant to diabetic keratoepitheliopathy. Cornea 2001; 20: 798- 801. 21. Gekka M, Miyata K, Nagai Y, et al. Corneal epithelial barrier function in diabetic patients. Cornea 2004; 23: 35- 7. 22. Abrams DA, Robin AL, Pollack IP, et al. The safety and efficacy of topical 1% alo 2145 ( p- aminoclonidine hydrochloride) in normal volunteers. Arch Ophthalmol 1987; 105: 1205- 7. 23. Jampel HD, Robin AL, Quigley HA, et al. Apraclonidine. a one- week dose- response study. Arch Ophthalmol 1988; 106: 1069- 72. 24. Robin AL. Short- term effects of unilateral 1% apraclonidine therapy. Arch Ophthalmol 1988; 106: 912- 5. 25. Stewart WC, Laibovitz R, Horwitz B, et al. A 90- day study of the efficacy and side effects of 0.25% and 0.5% apraclonidine vs 0.5% timolol. Arch Ophthalmol 1996; 114: 938^ 12. 26. Korczyn AD, Laor N, Nemet P. Sympathetic pupillary tone in old age. Arch Ophthalmol 1976; 94: 1905- 6. 27. Clark CV Autonomic neuropathy in proliferative diabetic retinopathy. Eye 1987; 1: 496- 9. 28. Smith SE, Smith SA, Brown PM. Cardiac autonomic dysfunction in patients with diabetic retinopathy. Diabetologia 1981; 21: 525- 8. 29 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. |