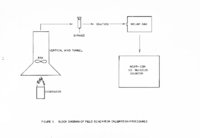
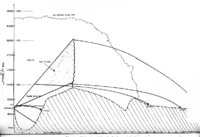
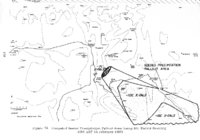
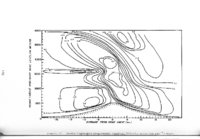
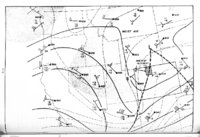
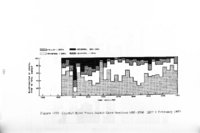
| OCR Text |
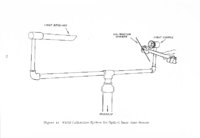
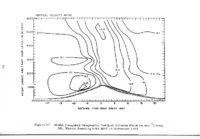
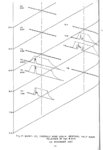
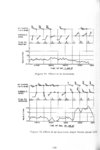
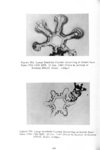
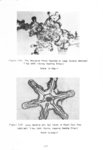
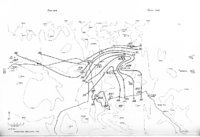
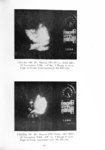
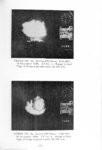
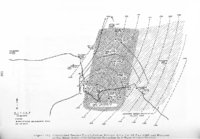
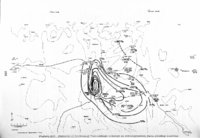
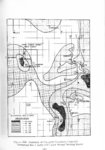
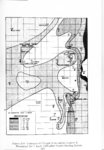
Show t i nated by Koenig (1959) and modified in the EG&G laboratory was used to identify Agl particles which had behaved as ice nuclei in collected snow samples. However, this technique required skillful, tedious, and expensive operations, which were not practical for large scale field experiments. Consequently, a more simple, inexpensive method was developed in the EG&G laboratory, and at about the same time by Knight (1969), and used during the 1968-69 season with good results. This new technique was based on the simple principle that any submicron size particle can act as a seed in its own supersaturated solution and can thus grow to any desired size. 1 I Using this principle, snow crystal replica slides collected in the Park Range Project Area were partially immersed in a supersaturated Agl-KI aqueous solution for 2 to 3 hours, and then allow e d to dry. Crystal photographs taken prior to immersion and drying were compared to photographs taken after tr e atment, and the percentage of individual crystal replicas which grew microscopically visible Agl crystals at the replica centers was computed. Examples of the visual appearance of crystal replicas with Agl centers are shown in Figures 9 and 10. Evidence of Agl scavenged on dendrite arms is visible in Figure 11. 2. 8 Silver Content of Snow Water An atomic absorption spectrophotometer has been used to analyze collected snow samples for the past two seasons. The methodology is described in detail by Parungo and Robertson (1969). Analysis for silver concentration \ in precipitation was performed by this technique using 1000 snow samples from the 1967 -68 operational season and 321 samples from the 1968-69 season. Initially, snow samples were taken from snow boards and through the snowpack after periods on the order of a week. Though some success was achieved, the primary fact learned was that it was more fruitful to add time resolution by taking individual shorter time period samples through a precipitation period, rather than taking one sample at the end of a long precipitation period. This proved more fruitful for two reasons: ( 1) If the release time is short compared to the total precipitation duration, the sample is likely to be diluted below the practical threshold of analysis . (2) Transport and dispersion characteristics change over a short time period so that the desired, or significant, results during a short time period may be diluted or masked by negative or indeterminate results during some other time period. 23 |