| OCR Text |
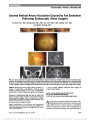
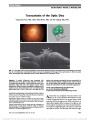
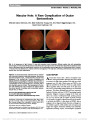
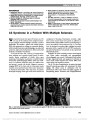
Show Transient Optic Perineuritis as the Initial Presentation of Central Nervous System Involvement by Pre-B Cell Lymphocytic Leukemia Justin H. Townsend, MD, Sander R. Dubovy, MD, Joshua Pasol, MD, Byron L. Lam, MD Abstract: A 20-year-old man with a history of pre-B cell acute lymphocytic leukemia (ALL) presented with optic perineuritis of the right eye while undergoing chemotherapy. Evaluation failed to reveal an infectious or neoplastic cause, and the patient improved with oral corticosteroid treatment. He returned 10 weeks later with complete loss of vision in the right eye. Optic nerve biopsy revealed leukemic infiltra-tion of the optic nerve, and the patient was treated for central nervous system (CNS) relapse of ALL. Transient optic perineuritis may be the initial manifestation of CNS involvement of pre-B cell ALL. Journal of Neuro-Ophthalmology 2013;33:162-164 doi: 10.1097/WNO.0b013e318281b84d © 2013 by North American Neuro-Ophthalmology Society Modern chemotherapeutic modalities for leukemia have dramatically increased survival with resultant increase in recurrence rates and metastatic disease. Optic perineuritis results from inflammation predominantly affect-ing the optic nerve sheath (1). The clinical presentation is similar to optic neuritis, with decreased vision and pain with eye movement. Symptoms are usually less severe than optic neuritis and transient in duration. Previous reports have associated optic perineuritis with inflammatory, infectious, and toxic causes (2,3). We report a patient who underwent systemic and prophylactic intrathecal chemotherapy for pre-B cell acute lymphocytic leukemia (ALL) and presented with a transient optic perineuritis that subsequently pro-gressed to infiltrative optic neuropathy with central retinal artery occlusion (CRAO). CASE REPORT A 20-year-old man with a 9-month history of pre-B cell ALL complained of right eye pain. He had received 5 cycles of chemotherapy using a Hyper-CVAD protocol, consisting of cyclophosphamide, vincristine, doxorubicin, dexamethasone courses alternated with methotrexate and cytarabine. There was no prior history or evidence of ALL central nervous system (CNS) involvement throughout his clinical course and per protocol had received 2 cycles of prophylactic intrathecal chemotherapy. A recent bone marrow biopsy done 2 months previously had shown no evidence of active leukemia. The visual acuity was 20/20 in each eye, with no relative afferent pupillary defect, full ocular motility but mild right eye pain on right gaze, and right optic disc edema. The patient reported light brightness of 90% in the right eye compared to the left. Automated visual field testing revealed only an enlarged blind spot of the right eye. Magnetic resonance imaging (MRI) demonstrated enhancement of the right orbital optic nerve sheath and possible thickening of the optic nerve (Fig. 1A). He was diagnosed with optic perineuritis. Cerebrospinal fluid (CSF) analysis was normal and negative for neoplastic cells and infectious agents. Rapid plasma regain, blood cultures, and purified protein derivative skin test and chest radiography were all unremarkable. The patient's upcoming cycle of chemotherapy was temporarily held, and he was treated with 60 mg of oral prednisone daily. His symptoms rapidly improved, with the ocular pain resolv-ing in 2 days, followed by resolution of optic disc edema and asymmetric brightness perception in 4 days. The visual acuity remained stable at 20/20 in each eye. He then underwent his sixth cycle of Hyper-CVAD and received an additional dose of intrathecal methotrexate, cytarabine, and hydrocortisone. The patient did not keep his follow-up appointment. Without our knowledge, he presented to an outside emergency room with chest pain 3 weeks later. He was found to be pancytopenic. Chest computed tomography Department of Ophthalmology, Emory Eye Center (JHT), Emory University, Atlanta, Georgia; and the Department of Ophthalmology, Bascom Palmer Eye Institute (SRD, JP, BLL), University of Miami Miller School of Medicine, Miami, Florida. The authors report no conflicts of interest. Address correspondence to Byron L. Lam, MD, Bascom Palmer Eye Institute, 900 NW 17th Street, Miami, Florida 33136; E-mail: blam@ med.miami.edu 162 Townsend et al: J Neuro-Ophthalmol 2013; 33: 162-164 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. (CT) revealed pulmonary nodules, and serum analysis for antibodies to aspergillus was negative. Ten weeks later, the patient reported a 2-day history of acute visual loss in the right eye. He had no light perception and funduscopy showed findings of a CRAO and venous stasis (Fig. 2A). Fluorescein angiography confirmed absent retinal arterial perfusion in the right eye. Repeat MRI showed persistent right optic nerve sheath enhancement and thickening of the optic nerve (Fig. 1B). Four days later, flocculent material was found protruding from the right optic nerve head (Fig. 2B), and the patient was diagnosed with an infiltrative optic neuropathy. Malignant cells were not found in CSF and bone marrow. Vitreous biopsy includ-ing material from the surface of the right optic disc was inconclusive, and cultures were negative. Right optic nerve biopsy revealed a monomorphic infiltrate and B cell mono-clonal population on flow cytometry (Fig. 3). The patient received radiation therapy to the brain and both orbits, high-dose intrathecal chemotherapy, and systemic chemotherapy. Repeat CSF analysis 2 months later revealed malignant cells. The patient's subsequent course was complicated by bacterial meningitis, Rhodotorula mucilaginosa sepsis and right-sided sinus mucormycosis with orbital involvement. He eventually achieved complete CNS and systemic remis-sion and received a matched, unrelated donor bone marrow transplant. At the last follow-up, 19 months after initial presentation, his vision remained no light perception, right eye, with optic disc pallor and attenuated retinal vessels. The left eye retains visual acuity of 20/20 with a normal ocular examination. DISCUSSION Optic perineuritis is an inflammatory optic neuropathy with symptoms similar to those of optic neuritis including pain with eye movement, variably decreased visual acuity and optic disc edema (4). Optic perineuritis has distinct neuro-imaging features compared to optic neuritis, specifically pre-dominant enhancement of the optic nerve sheath and perineural fat stranding rather than enhancement of the optic nerve itself (1). Following intravenous contrast, there is a "tram track" appearance on axial views, or a "doughnut" on coronal views, at times resembling optic nerve sheath meningioma. Compared to optic neuritis, visual loss is less profound in optic perineuritis with .50% of patients retain-ing acuity of at least 20/20 (4). Perineuritis is usually slowly progressive, and patients typically have a good response to oral corticosteroids although recurrence upon tapering of steroids has been reported. There is no increased risk of multiple sclerosis in patients with optic perineuritis (1). FIG. 1. Contrast-enhanced T1 coronal magnetic resonance imaging (MRI) with fat suppression A. At presentation, there is enhancement of the right optic nerve sheath and perineural fat stranding. B. Ten weeks later, MRI shows enhancement of the right optic nerve sheath and optic nerve thickening. FIG. 2. A. Right fundus 2 days after the acute loss of vision showing central retinal artery occlusion, venous stasis, and optic disc edema. B. Four days after acute loss of vision, there is peripapillary infiltrate and flocculent material protruding from the right optic nerve head into the vitreous cavity. Townsend et al: J Neuro-Ophthalmol 2013; 33: 162-164 163 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. A wide spectrum of disorders are associated with optic perineuritis, including inflammatory (idiopathic orbital inflammation, Wegener granulomatosis, sarcoidosis), infec-tious (herpesviridae, mycobacterium, syphilis, Toxocara, HIV [1,2]), and toxic (imatinib, methotrexate, cytarabine, linezolid [2,3]). Our patient presented with optic perineuritis as the initial manifestation of leukemic invasion of the CNS. After initial recovery, he lost vision due to leukemic infiltration of the optic nerve with venous stasis. Infiltrative optic neuropathy causes visual loss through progressive, compressive necrosis, or ischemia of the optic nerve (5). Multiple neoplastic processes have been reported to manifest as an infiltrative optic neuropathy, including leukemia, lymphoma, optic nerve glioma, meningioma, and metastatic malignancy (6,7). The optic nerve and peri-neural structures are an infrequent yet important site of CNS leukemic involvement. Active bone marrow and sys-temic disease is typically present with leukemic infiltration of the optic nerve (8,9). However, there are reports of optic nerve involvement during systemic remission, as in our case (10,11). An infectious optic neuropathy also must be considered in patients undergoing chemotherapy, who are at an increased risk for opportunistic infections. Sino-orbital aspergillosis and rhino-orbital-cerebral mucomycosis are 2 disorders of particular concern in the immunocompromised patient (12). In our patient, the rapidity of the infiltrative process and high concern for opportunistic infection led to aggressive diagnostic evaluation and treatment. After vitreous biopsy was nondiagnostic, we proceeded to biopsy the optic nerve in order to differentiate leukemic disease from opportunistic infection (13). Optic nerve biopsy was definitive for neoplastic involve-ment. Other diagnostic modalities including CSF and bone marrow analysis remained noncontributory for an addi-tional 2 months. By that time, the patient had received intrathecal and systemic chemotherapy and radiation therapy (7,10,14,15). His fellow optic nerve remained free of leukemic involvement at the conclusion of his treatment course, achieving CNS and systemic remission. REFERENCES 1. Fay AM, Kane SA, Kazim M, Millar WS, Odel JG. Magnetic resonance imaging of optic perineuritis. J Neuroophthalmol. 1997;17:247-249. 2. Winterkorn JM, Odel JG, Behrens MM, Hilal S. Large optic nerve with central retinal artery and vein occlusions from optic neuritis/perineuritis rather than tumor. J Neuroophthalmol. 1994;14:157-159. 3. de Fátima Soares M, Braga FT, da Rocha AJ, Lederman HM. Optic nerve infiltration by acute lymphoblastic leukemia: MRI contribution. Pediatr Radiol. 2005;35:799-802. 4. Purvin V, Kawasaki A, Jacobsen D. Optic perineuritis: clinical and radiographic features. Arch Ophthalmol. 2001;119:1299- 1306. 5. Ellis W, Little HL. Leukemic infiltration of the optic nerve head. Am J Ophthalmol. 1973;75:867-871. 6. Nikaido H, Mishima H, Ono H, Choshi K, Dohy H. Leukemic involvement of the optic nerve. Am J Ophthalmol. 1988;105:294-298. 7. Takahashi T, Oda Y, Isayama Y. Leukemic optic neuropathy. Ophthalmologica. 1982;185:37-45. 8. Kincaid MC, Green WR. Ocular and orbital involvement in leukemia. Surv Ophthalmol. 1983;27:211-232. 9. Rosenthal AR. Ocular manifestations of leukemia. Ophthalmology. 1983;90:899-905. 10. Sharma T, Grewal J, Gupta S, Murray PI. Ophthalmic manifestations of acute leukaemias: the ophthalmologist's role. Eye. 2004;18:663-672. 11. Ohkoshi K, Tsiaras WG. Prognostic importance of ophthalmic manifestations in childhood leukaemia. Br J Ophthalmol. 1992;76:651-655. 12. Choi MY, Bae IH, Lee JH, Lee SJ. Aspergillosis presenting as an optic neuritis. Korean J Ophthalmol. 2002;16:119-123. 13. Wolfe GI, Galetta SL, Mollman JE. Spontaneous remission of papilledema and sixth nerve palsy in acute lymphoblastic leukemia. J Neuroophthalmol. 1994;14:91-94. 14. Ha CS, Chung WK, Koller CA, Cox JD. Role of radiation therapy to the brain in leukemic patients with cranial nerve palsies in the absence of radiological findings. Leuk Lymphoma. 1999;32:497-503. 15. Reddy SC, Menon BS. A prospective study of ocular manifestations in childhood acute leukaemia. Acta Ophthalmol Scand. 1998;76:700-703. FIG. 3. Right optic nerve biopsy. A. Basophilic infiltrate is present in the substance of the nerve (hematoxylin & eosin x2). B. Dense monomorphic cellular infiltrate within the substance of the nerve with surrounding areas of necrosis (hematoxylin & eosin ·20). C. CD79 immunostain shows diffuse positivity indicating predominant pre-B-cell population (·20). 164 Townsend et al: J Neuro-Ophthalmol 2013; 33: 162-164 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |