| OCR Text |
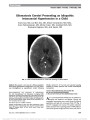
Show Cortical Blindness Following a Near-Drowning Incident Ellen H. Koo, MD, Jerrold L. Boxerman, MD, PhD, Marjorie A. Murphy, MD Abstract: Victims of near-drowning incidents often suffer neurologic injury with long-term sequelae secondary to hypoxic-ischemic injury. We describe a case of profound visual loss due to bilateral occipital lobe infarcts in a 23- year-old male victim of a near-drowning incident. Journal of Neuro-Ophthalmology 2011;31:347-349 doi: 10.1097/WNO.0b013e318228dc87 2011 by North American Neuro-Ophthalmology Society Near-drowning is a term applied when an individual survives for at least some period after suffocation from submersion in a liquid (1). The primary mechanism of injury is pulmonary failure caused by fluid aspiration, which results in severe arterial hypoxemia and secondary ischemic damage to other organs, including the brain (2). Victims of near-drowning incidents often suffer neurologic injury with long-term sequelae secondary to hypoxic-ischemic injury (3). We describe a case of cortical blindness due to bilateral occipital lobe infarcts following a near-drowning incident. A 23-year-old man was found unresponsive at the beach after a near-drowning incident while surfing. He was in-tubated and admitted to the Trauma Intensive Care Unit. CT of the chest showed bilateral airspace disease consistent with aspiration. MRI of the cervical spine showed a fracture of the C-5 vertebral body that did not require surgical intervention. Shortly after the patient was extubated on the fourth day of his hospitalization, he complained of profound bilateral vision loss. He was awake, alert, and cooperative with the examination. Visual acuity was counting fingers in both eyes, and ophthalmologic examination was otherwise nor-mal. CT of the brain was unremarkable while MRI with FLAIR sequences revealed widespread areas of abnormal hyperintensity (Fig. 1, top panel). Diffusion-weighted im-ages (DWI) confirmed the presence of restricted diffusion in the affected regions (Fig. 1, middle panel), and these changes were consistent with cytotoxic edema (Fig. 1, bottom panel). There was no improvement in vision upon his discharge from the hospital 6 days later. The patient refused further follow-up. The causes of cortical blindness may be broadly divided into vascular, toxic, traumatic, infectious, and neurode-generative. Near-drowning leads to severe arterial hypox-emia and secondary ischemic infarcts of the brain (2) and is a potential vascular source of injury leading to cortical blindness. During a drowning or a near-drowning event, cardio-pulmonary failure from aspiration causes decrease in cere-bral blood flow leading to ischemic injury (2). The areas of greatest susceptibility to ischemic injury are usually the vascular end zones (‘‘watershed'' areas), as well as the hip-pocampus, insular cortex, and basal ganglia (4). However, with greater severity of hypoxic ischemia, more extensive and global neocortical damage can occur (4). Posterior reversible encephalopathy syndrome (PRES) is another vascular etiology of cortical blindness (5,6). This seems unlikely in our patient. First, PRES is typically precipitated by sudden hypertension and is associated with headache, seizure, visual disturbance, and altered mental function (6). In cases of near-drowning, the mechanism of injury to the brain is as-sociated with hypotension and hypoxia (7). Second, the lesions of PRES on DWI are usually isointense or hypointense, with areas of increased signal on apparent diffusion coefficient (ADC) maps indicating vasogenic edema (5). In our patient, the results of DWI indicated cytotoxic edema. Third, PRES is usually reversible with a favorable visual outcome while our patient remained severely visually impaired. Following cold-water submersion, protecting the brain from hypoxic injury can be favorably modified by the co-existence of hypothermia (8). Cold water is considered to be neuroprotective in near-drowning due to cold-induced Departments of Ophthalmology (EHK, MAM) and Diagnostic Im-aging (JLB), The Warren Alpert Medical School of Brown University, Rhode Island Hospital, Providence, Rhode Island. Presented as poster at the 36th North American Neuro-Ophthal-mology Society Annual Meeting, March 9, 2010, Tucson, AZ. The authors report no conflicts of interest. Address correspondence to Marjorie A. Murphy, MD, Department of Ophthalmology, Rhode Island Hospital Coro Center, Suite 202, 1 Hoppin Street, Providence, RI 02903; E-mail: margiemurphy@ cox.net Koo et al: J Neuro-Ophthalmol 2011; 31: 347-349 347 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. decrease in cerebral metabolic rate for oxygen (9,10). In animal studies, brain temperature below 85 F is thought to provide an increased protection against anoxia (11), and this appears to be the case in humans as well (12). The water in which our patient was found ranged in temperature from 68.71 to 71.89 F. It is possible that this cold-water FIG. 1. Top panel: Axial FLAIR images demonstrating abnormal hyperintense signal (arrowheads) in the frontoparietal (A) parieto-occipital (B), and temporo-occipital (C) cortex. There is also involvement of the caudate nuclei (arrows) (B). Middle panel: Axial DWI scans reveal bilateral scattered regions of abnormal gyriform signal hyperintensity corre-sponding to regions of signal abnormality detected on FLAIR images. Bottom panel: In areas of DWI hyperintensity, apparent diffusion coefficient (ADC) maps showing reduced signal consistent with cytotoxic edema. 348 Koo et al: J Neuro-Ophthalmol 2011; 31: 347-349 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. submersion allowed some degree of neuroprotection, thereby leading to focal infarcts rather than diffuse cerebral involvement. REFERENCES 1. Neal JM. Near-drowning. J Emerg Med. 1985;3:41-52. 2. Hasibeder W, Friesenecker B, Mayr A. Near drowning: epidemiology-pathophysiology-therapy. Anasthesiol Intensivmed Notfallmed Schmerzther. 2003;38:333-340. 3. Sachdeva RC. Near drowning. Crit Care Clin. 1999;15: 281-296. 4. Ibsen LM, Koch T. Submersion and asphyxia injury. Crit Care Med. 2002;30:S402-S408. 5. Lamy C, Oppenheim C, Me´der JF, Mas JL. Neuroimaging in posterior reversible encephalopathy syndrome. J Neuroimaging. 2004;14:89-96. 6. Kahana A, Rowley HA, Weinstein JM. Cortical blindness: clinical and radiologic findings in reversible posterior leukoencephalopathy syndrome: case report and review of the literature. Ophthalmology. 2005;112:7-11. 7. Murray RR, Kapila A, Blanco E, Kagan-Hallet KS. Cerebral computed tomography in drowning victims. AJNR Am J Neuroradiol. 1984;5:177-179. 8. Elixson EM. Hypothermia. Cold water drowning. Crit Care Nurs Clin North Am. 1991;3:287-292. 9. Ginsberg MD, Globus MY, Dietrich WD, Busto R. Temperature modulation of ischemic brain injury: a synthesis of recent advances. Prog Brain Res. 1993;96: 13-22. 10. Michenfelder JD, Milde JH. The relationship among canine brain temperature, metabolism, and function during hypothermia. Anesthesiology. 1991;75:130-136. 11. Beetar JT. Hypoxia after near drowning. In: Morgan JE, Baron IS, Ricker JH, eds. Casebook of Clinical Neuropsychology. New York, NY: Psychology Press, 2009: 255. 12. Orlowski JP. Drowning, near-drowning, and ice-water submersions. Pediatr Clin North Am. 1987;34:75-92. Koo et al: J Neuro-Ophthalmol 2011; 31: 347-349 349 Clinical Observation Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |