| OCR Text |
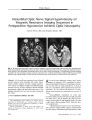
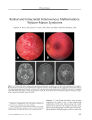
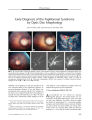
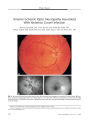
Show ORIGINAL CONTRIBUTION Necrobiotic Xanthogranuloma Associated with Choroidal Infiltration and Syncytial Giant Cell Hepatitis Radgonde Amer, MD, Jacob Pe ' er, MD, Orit Pappo, MD, and Shlomo Dotan, MD Abstract: A 31- year- old woman developed necrobiotic xanthogranuloma ( NXG), a thickened choroid, and syncytial giant cell hepatitis, a previously unreported association. NXG and syncytial giant cell hepatitis may have a common autoimmune pathogenesis. ( J Neuro- Ophthalmol 2005; 25: 189- 192) Necrobiotic xanthogranuloma ( NXG) is a rare non- Langerhans cell histiocytosis characterized by cutaneous, visceral, and ophthalmologic lesions and sometimes monoclonal gammopathy and lymphoproliferative diseases ( 1- 5). Cutaneous involvement is characterized by the development of multiple xanthomatous plaques with prominent telangiectasia and bulky subcutaneous nodules most commonly found in the periorbital area, trunk, and extremities ( 3,5). Visceral involvement may include the lungs, heart, liver, and spleen. Ophthalmologic involvement includes conjunctival hyperemia, keratitis, episcleral plaques, episcleritis, scleritis, anterior uveitis, and infiltrative orbital masses with extraocular muscle and lacrimal gland involvement ( 2,5). The accompanying paraproteinemia tends to be a kappa or lambda IgG monoclonal gammopathy. Neutropenia, hypocomplementemia, cryoglobulinemia, and hy-perlipidemia are additional features. Cases with monoclonal gammopathy are at risk for developing multiple myeloma, macroglobulinemia, amyloidosis, or malignant lymphoproliferative diseases ( 4,5). Treatment consists of local excision, systemic corticosteroids, chemotherapy, and plasmapheresis. We describe a previously unreported association of the rare syncytial giant cell hepatitis with NXG and choroidal thickening. Departments of Ophthalmology ( RA, JP, SD) and Pathology ( OP), Hadassah- Hebrew University Medical Center, Jerusalem, Israel. Address correspondence to Radgonde Amer, MD, Department of Ophthalmology, Hadassah University Hospital, P. O. Box 12000, Jerusalem 91120, Israel; E- mail: radgondeamer@ yahoo. com CASE REPORT A 31- year- old woman presented to Hadassah University Hospital, Jerusalem, Israel, with arthralgia and arthritis of the small and large joints. She also experienced generalized fatigue, weight loss, ecchymosis in her lower extremities, and erythematous rash over the affected joints. She had hepatosplenomegaly; there were no neurologic deficits. Laboratory investigations revealed an elevated erythrocyte sedimentation rate of 80 mm/ h and elevated hepatocellular and cholestatic liver enzymes ( three times the upper limit of normal). Synthetic liver functions, including coagulation factors, were normal. Imaging studies, including abdominal ultrasound and computed tomography, revealed splenomegaly of 17 cm and hepatomegaly of 18 cm. There were no signs of lymphadenopathy in cervical, mediastinal, or abdominal sites. There was no evidence of Wilson's disease. A virologic survey for any signs of current liver infection resulting from hepatofrophic viruses was negative for hepatitis viruses A, B, and C. She had IgG antibodies to hepatitis A virus, indicating a past infection. Liver biopsy revealed syncytial giant cell hepatitis ( Fig. 1). However, there were no indications in the biopsy or in the serum of paramyxovirus, reducing the possibility of an association with paramyxovirus infection ( 6). Additional investigations revealed a paraproteinemia of IgG- kappa with elevated IgM and IgG and a normal level of IgA. C3 levels were below the lower limit of normal; anti- mitochondrial, anti- smooth muscle, and ANA were negative. Bone marrow biopsy was normal. A treatment trial with prednisone ( 1 mg/ kg/ day) and azathioprine ( 2 mg/ kg/ day) was instituted. Neither agent markedly affected her disease either clinically or histologically, as revealed by a second liver biopsy performed several months later. One year later, she presented with bilateral conjunctival injection and corneal annular subepithelial infiltrates. She was treated with topical corticosteroids with good response. Nine months later, she reported a mild decrease in vision in the OS. Visual acuity was 20/ 20 OD and 20/ 30 OS. Anterior segment examination showed the corneal J Neuro- Ophthalmol, Vol. 25, No. 3, 2005 189 J Neuro- Ophthalmol, Vol. 25, No. 3, 2005 Amer et al FIG. 1. Liver biopsy shows chronic portal inflammation with focal interface hepatitis. Several multinucleated giant hepatocytes are noted in the parenchyma ( hematoxylin & eosin X400). findings previously described. The fundus appeared normal OD; a thickened choroid was seen nasal to the optic disc OS. Echography showed thickening of the choroid in both eyes of 4.5 mm ( Fig. 2). Treatment with 1.5 mg/ kg prednisone per day accomplished complete clinical and ultrasonic resolution in both eyes over three weeks. Her subsequent clinical course was characterized by recurrent attacks approximately once per year of visual deterioration accompanied by choroidal thickening. Response to systemic corticosteroids soon became partial with a residual visual acuity of 20/ 40 OU. Cyclosporine A was introduced at a dose of 4 mg/ kg per day, but the case negligently discontinued it a few months later. FIG. 2. B- scan echography illustrates thickening of the choroid OS ( arrow). Four years after the initial ophthalmologic evaluation, she was found to have bilateral periorbital plaques extending in a band- like fashion in both upper lids. Shortly thereafter, she developed binocular vision loss with visual acuities of finger counting OD and 20/ 200 OS. Intraocular pressure was normal bilaterally. Episcleritis and mild iridocyclitis were noted OU. Marked choroidal thickening was noted OU with congestion of the optic discs bilaterally. Echography showed increased thickness of the choroid in both eyes to 6.5 mm. Thickening of the extraocular muscles was seen on magnetic resonance imaging. Prednisone at a dosage of 1.5 mg/ kg per day was initiated with partial response. Despite treatment with 20 mg subconjunctival methylprednisolone acetate, visual acuity did not improve OD. Radial macular folds were noted OD ( Fig. 3). Visual acuity OS improved to 20/ 50. On corticosteroid tapering, visual acuity fell to hand movements OD and finger counting OS. Ophthalmoscopy showed optic disc swelling with macular edema bilaterally. Intravenous pulse methylprednisolone at a dosage of 1 g per day and 3 mg/ kg cyclosporine A per day were administered. Biopsy of the periorbital plaques disclosed a distinctive pattern of subepidermal granulomatous masses with focal aggregates of histiocytes and giant cells surrounded by hyaline necrobiosis, compatible with a diagnosis of necrobiotic xanthogranuloma ( Fig. 4). Bone marrow biopsy was normal. An audiogram revealed moderate sensorineural loss at 8000 Hz in the left ear. She had hypercholesterolemia. After a 10- year period under our care, the case's visual acuity was 20/ 60 OD and 20/ 200 OS with a radial macular- fold OD and cystoid macular edema OS. Both optic discs had blurred margins. No evidence of lympho-proliferative or myeloproliferative disease has appeared. 190 © 2005 Lippincott Williams & Wilkins Necrobiotic Xanthogranuloma J Neuro- Ophthalmol, Vol. 25, No. 3, 2005 FIG. 4 . A. Biopsy of the eyelid lesion shows mononucleated and multinucleated histiocytes and lymphocytes surrounding an area of hyaline necrobiosis ( hematoxylin & eosin, X100). B. High magnification of the same lesion shows hyaline necrobiosis above and mononucleated and multinucleated histiocytes and lymphocytes below ( hematoxylin & eosin, X400). DISCUSSION Our case represents a unique case in harboring two rare diseases: NXG and syncytial giant cell hepatitis. Winkelmann et al ( 7) reported the occurrence of giant cell granulomatous pulmonary and myocardial lesions in NXG with paraproteinemia. Umbert and Winkelmann ( 8) reported the occurrence of xanthogranuloma of the spleen with giant cell myocarditis in one case. Two case reports described liver involvement in NXG, one with liver amyloidosis ( 9) and the other with nodular transformation ( 10). Our case had giant cell hepatitis proven by biopsy and also had splenomegaly of unidentified etiology, but according to the previous reports, possible pathogenetic mechanisms should include giant cell granulomas and xanthogranulomas. Syncytial giant cell hepatitis is an uncommon form of hepatic inflammation characterized by giant cell transformation of hepatocytes. This peculiar reaction pattern does not appear to represent a specific disease and has been associated with various etiologies, including drug- taking, autoimmune diseases, and viral infection. The hepatitis may evolve to chronic cholestatic cirrhosis or it may lead arapidly progressive course, ending in fulminant liver failure necessitating liver transplantation ( 11). Phillips et al ( 6) described intracytoplasmic structures consistent with para-myxoviral nucleocapsids. This finding has now been reported elsewhere ( 12), and others have reported the efficacy of ribavirin for the treatment of giant cell hepatitis, providing evidence for its viral origin ( 13). Despite the unfavorable prognosis in the majority of cases described, our case had a rather stable condition and non- progressive histologic features on repeated liver biopsies. Immunosuppressive agents did not alter the clinical course, and liver transplantation has not been necessary. Choroidal thickening has not been reported in NXG. It showed a limited response to systemic corticosteroids or cyclosporine A. Biopsy of the periorbital plaques had the only decisive role in making the diagnosis. Such plaques are known to occur in the majority of cases of NXG ( 2). Cogan syndrome was also present in our case, because she had bilateral corneal subepithelial infiltrates and was discovered 6 years after her first presentation to have moderate sensorineural loss in her left ear. This auditory dysfunction is speculated to have developed much earlier but had been clinically masked as a result of the systemic treatment with corticosteroids. The association between Cogan syndrome and NXG was previously reported by Cornblath et al ( 14). Their case's hearing loss was unresponsive to corticosteroids and eventually required a cochlear implant. The relationship of NXG to syncytial giant cell hepatitis remains speculative. The occurrence of the two conditions in a single case could be a coincidence or a reflection of a common etiology. Autoimmunity is a mechanism that has been applied to syncytial giant cell hepatitis and to Cogan syndrome. The main concern in necrobiotic xanthogranuloma is a 15% to 20% increased risk for development of myeloproliferative disease ( 3). This association emphasizes the need for life- long follow up and repeated bone marrow biopsies. Various hypotheses have been proposed to explain the pathophysiology of NXG. It has been suggested that the paraprotein in NXG has functional features of a lipoprotein that may bind to lipoprotein receptors of histiocytes and induce granuloma formation ( 15). It has also been proposed that the paraproteinemia is the primary phenomenon with secondary proliferation of macrophages containing receptors for the Fc portion of IgG ( 16). REFERENCES Kossard S, Winkelmann RK. Necrobiotic xanthogranuloma with paraproteinemia. J Am Acad Dermatol 1980; 3: 257- 70. Robertson DM, Winkelmann RK. Ophthalmic features of necrobiotic xanthogranuloma with paraproteinemia. Am J Ophthalmol 1984; 97: 173- 8. Finan MC, Winkelmann RK. Necrobiotic xanthogranuloma with paraproteinemia. A review of 22 cases. Medicine 1986; 65: 376- 88. Venencie PY, Puissant A, Verola O, et al. Necrobiotic xanthogranuloma with myeloma. A case report. Cancer 1987; 59: 588- 92. Ugurlu S, Bartley GB, Gibson LE. Necrobiotic xanthogranuloma: Long- term outcome of ocular and systemic involvement. Am J Ophthalmol 2000; 129: 651- 7. Phillips MJ, Blendis LM, Poucell S, et al. Syncytial giant- cell hepatitis. Sporadic hepatitis with distinctive pathological features, 191 J Neuro- Ophthalmol, Vol. 25, No. 3, 2005 Amer et al a severe clinical course, and paramyxoviral features. N Engl J Med 1991; 324: 455- 60. 7. Winkelmann RK, Litzow MR, Umbert IJ, et al. Giant cell granulomatous pulmonary and myocardial lesions in necrobiotic xanthogranuloma with paraproteinemia. Mayo Clin Proc 1997; 72: 1028- 33. 8. Umbert I, Winkelmann RK. Necrobiotic xanthogranuloma with cardiac involvement. Br J Dermatol 1995; 133: 438^ 3. 9. Westermann G, August C, Bonsmann G, et al. [ Necrobiotic xanthogranuloma with skin and liver amyloidosis.] Med Klin 2001; 96: 50^ k 10. Novak PM, Robbins TO, Winkelmann RK. Necrobiotic xanthogranuloma with myocardial lesions and nodular transformation of the liver. Hum Pathol 1992; 23: 195- 6. 11. Lau JY, Koukoulis G, Mieli- Vergani G, et al. Syncytial giant- cell hepatitis- A specific disease entity? J Hepatol 1992; 15: 216- 9. 12. Durand F, Degott C, Sauvanet A, et al. Subfulminant syncytial giant cell hepatitis: Recurrence after liver transplantation treated with ribavirin. J Hepatol 1997; 26: 722- 6. 13. Hassoun Z, N'Guyen B, Cote J, et al. A case of giant cell hepatitis recurring after liver transplantation and treated with ribavirin. Can J Gastroenterol 2000; 14: 729- 31. 14. Cornblath WT, Dotan SA, Trobe JD, et al. Varied clinical spectrum of necrobiotic xanthogranuloma. Ophthalmology 1992; 99: 103- 7. 15. Char DH, LeBoit PE, Ljung BM, et al. Radiation therapy for ocular necrobiotic xanthogranuloma [ Letter]. Arch Ophthalmol 1987; 105: 174- 5. 16. Rappersberger K, Wrba F, Heinz R, et al. [ Necrobiotic xanthogranuloma in paraproteinemia.] [ German] Hautarzt 1989; 40: 358- 63. 192 © 2005 Lippincott Williams & Wilkins |