| OCR Text |
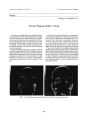
Show Journal of Neuro- Ophthalmology 17( 4): 262- 265, 1997. © 1997 Lippincott- Raven Publishers, Philadelphia J Complete Ophthalmoplegia After Zoster Ophthalmicus Anne Chang- Godinich, M. D., Andrew G. Lee, M. D., Paul W. Brazis, M. D., Thomas J. Liesegang, M. D., and Dan B. Jones, M. D. Complete ophthalmoplegia following herpes zoster ophthalmicus ( HZO) is rare. We report three cases of HZO- associated complete ophthalmoplegia and review thirteen additional cases reported in the English language medical literature over the past 30 years. HZO- associated complete ophthalmoplegia occurs mostly in individuals over the age of 50 years and usually occurs within one to two weeks of the development of cutaneous HZO. The prognosis for recovery is good, with significant improvement typically seen within 2 months and complete or near resolution within 18 months time. Key Words: Complete ophthalmoplegia- Herpes zoster ophthalmicus. Although ocular motor cranial neuropathy ( e. g., third, fourth, or sixth nerve palsy) may occur following cutaneous Herpes zoster ophthalmicus ( HZO), complete ophthalmoplegia is uncommon ( 1- 3). We report on three cases of total ophthalmoplegia after HZO and review the literature on the clinical manifestations and treatment for this disorder ( 3- 9). CASE REPORTS Case 1 An 82- year- old woman presented with a painful complete ophthalmoplegia OS. Her past medical history was significant for 25 years of rheumatoid arthritis, for which she was treated with chronic low- dose oral prednisone and intermittent pulse dose methotrexate. She had well-controlled hypertension and hypothyroidism. Her past ocular history was significant for long- standing bilateral visual loss resulting from age- related macular degeneration, and she had undergone cataract removal in both eyes. Her medications on admission were prednisone 4 Manuscript received December 11, 1996; accepted April 15, 1997. From the Departments of Ophthalmology ( A. C., A. G. L., D. B. J.), Neurology ( A. G. L.), and Neurosurgery ( A. G. L.), Baylor College of Medicine, Houston, Texas, and Departments of Ophthalmology ( P. W. B., T. J. L.) and Neurology ( P. W. B.), Mayo Clinic Jacksonville, Jacksonville, Florida, U. S. A., and the Division of Neurosurgery, University of Texas, M. D. Anderson Cancer Center, Houston, Texas, U. S. A. ( A. G. L.). Address correspondence to: Andrew G. Lee, M. D., Departments of Ophthalmology, Neurology, and Neurosurgery, Cullen Eye Institute, 6501 Fannin Street, NC- 200, Houston, Texas 77030 U. S. A. mg per day, nifedipine, thyroxine, estrogen, and medroxyprogesterone. She was well until September 1996, when she developed acute onset of pain and a vesicular cutaneous eruption in the first ( ophthalmic) division of the trigeminal nerve ( VI) on the left side of her face. She was admitted to the hospital, underwent a 3- day course of intravenous acyclovir, and was discharged on a 10- day course of oral acyclovir. One week following development of the skin rash, she was noted to have ptosis OS. An ophthalmologist noted the ptosis, but there were no pupil or extraocular motility abnormalities. One week later, she was noted to have inability to move her left eye and was referred to the Neuro- Ophthalmology Service ( A. G. L.) at Baylor College of Medicine. Ophthalmologic examination on October 4, 1996 revealed a best corrected visual acuity of 20/ 200 OD and 20/ 70 OS. The pupils measured 4 mm OD and 5 mm OS, with poor light reaction OD, and no light or near reaction OS. There was no afferent pupillary defect. She correctly identified 11 of 14 Ishihara color plates in each eye. External examination revealed complete ptosis OS. There were several healing vesicles in the VI distribution of the left trigeminal nerve. Corneal reflex OS was diminished, and she had pain and hypesthesia in the VI distribution of the left trigeminal nerve. Extraocular motility examination revealed a complete inability to ad-duct, elevate, or depress as well as partial inability to abduct the left eye ( Fig. 1). The right eye moved normally. Visual fields were full- to- confrontation. Slit- lamp biomicroscopy revealed a posterior chamber lens implant in each eye. There was no evidence of anterior segment inflammation. Intraocular pressures were normal OU. Ophthalmoscopy revealed macular drusen and geographic atrophy of the retinal pigment epithelium centrally OU, consistent with her visual acuity. Magnetic resonance ( MR) scans of the head and orbits were normal. Specifically, there was no abnormality in the cavernous sinus or orbit on the left. A lumbar puncture revealed an opening pressure of 11 cm of water ( normal, < 20 cm of water); protein, 68 mg/ 100 ml ( normal, 15- 50 mg/ ml); glucose, 61 mg/ 100 ml; 10 red blood cells; and 10 white cells with a differential of 90% lymphocytes. The patient was treated with intravenous acyclovir ( 1,200 mg/ day for 7 days), intravenous methyl-prednisolone ( 1,000 mg/ day for 5 days), and carbamaz- 262 COMPLETE OPHTHALMOPLEGIA AFTER ZOSTER OPHTHALMICUS 263 FIG. 1. Case 1. Extraocular motility series demonstrates a complete inability to adduct, elevate, or depress as well as partial inability to abduct the left eye. Arrows indicate direction of gaze. epine ( 400 mg/ day) for one month. She experienced moderate improvement in the severity of her facial pain and slow improvement of her ophthalmoplegia. One month later, the ptosis OS had resolved, she had no diplopia or pain, and extraocular motility had improved dramatically . Case 2 A 72- year- old woman presented with a painful ophthalmoplegia OS. Her past medical history was significant for 19 years of chronic lymphocytic leukemia ( CLL) treated with pulse chemotherapy. In October 1993, she developed a rash involving the left VI distribution of the trigeminal nerve and was treated with oral acyclovir for several days followed by a 6- day course of intravenous acyclovir. Her other medications at the time of presentation included ciprofloxacin, amitriptyline, and propoxyphene. Two weeks later, she developed ptosis, pain, and ophthalmoplegia OS. Ophthalmologic examination on November 29, 1993, at the Mayo Clinic- Jacksonville ( P. W. B.) revealed a visual acuity of 20/ 20 OD and 20/ 200 OS. Pupils measured 5 mm OD and 7 mm OS, with good light reaction OD and poor reaction OS. A left afferent pupillary defect was present. External examination revealed 6 mm of ptosis OS. The corneal reflex OS was diminished and she had sensory loss in the VI distribution of the left trigeminal nerve. The right eye moved normally, but there was nearly complete ophthalmoplegia of the left eye, with only minimal preservation of abduction. Slit- lamp biomicroscopy revealed mild conjunctival injection OS without evidence of anterior segment inflammation. Intraocular pressures and ophthalmoscopy were normal OU. Although the optic nerve appeared normal, she was believed to have a left optic neuropathy. MR scans of the head and orbits were normal. The patient was treated with oral acyclovir ( 1,600 mg/ day) for 3 months, and prednisone ( 60 mg/ day) for 8 weeks with a tapering dose. After 7 days, the ptosis, lid edema, and conjunctival injection improved, and visual acuity was 20/ 60 OS. After 4 weeks of treatment, visual acuity was 20/ 30 OS, the eye pain improved, and there was modest improvement of motility OS. Follow- up examination, on January 19, 1994, after 8 weeks of treatment revealed a visual acuity of 20/ 25 OS. The pupils measured 4 mm OD and 6 mm OS, with good light reaction OD and trace light reaction OS. No afferent pupillary defect was seen. External examination revealed 2 mm of residual ptosis OS. The corneal reflex OS remained diminished and sensory loss in the left VI cutaneous distribution persisted. Motility examination revealed an ex-otropia measuring 12 prism diopters ( PD) at distance, with a left hypotropia of 6 PD and only mild adduction and elevation deficits OS. The prednisone was tapered and the patient was treated with acyclovir for 3 months. Case 3 A 75- year- old man presented with painful decreased vision in his left eye. His past medical history was significant for hypertension and myocardial infarction 1 year earlier. He was taking naldol ( 40 mg/ day). He was well until the middle of November 1982, when he developed a painful vesicular rash in the VI distribution of the left trigeminal nerve. He was evaluated by an ophthalmologist, who treated him with topical prednisolone, atropine, and timolol in the left eye for 4 weeks. He was referred to Baylor College of Medicine ( D. B. J.) after development of ptosis and complete ophthalmoplegia OS and slurred speech. Ophthalmologic examination on December 16, 1982 revealed a best corrected visual acuity of 20/ 20 OD and 20/ 100 OS. The pupils measured 4 mm OD and 6.5mm OS, with no light or near reaction OS. There was no definite afferent pupillary defect. External examination revealed a resolving vesicular rash in the VI distribution of the left trigeminal nerve. There was an almost complete ptosis of the left eye with 4 mm of proptosis. Motility examination revealed a complete inability to elevate or depress and partial inability to adduct and ab- J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 264 A. CHANG- GODINICH ET AL. duct the left eye. The right eye moved normally. Visual field testing by Aimark projection perimetry revealed an inferior paracentral scotoma nasally OS. Slit- lamp biomicroscopy of the left eye revealed an irregular corneal epithelium, Descemet's membrane folds in the su-perotemporal periphery, mild anterior chamber reaction, and endothelial keratitic precipitates inferiorly. Intraocular pressure was 16 mm OD and 24 mm OS. Ophthalmoscopy was normal OU. Computed tomography ( CT) scan of the head and orbits showed mild proptosis of the left eye with soft tissue swelling anterior to the globe. The patient was continued on topical medications, with tapering of the prednisolone. Five weeks later, the ptosis had improved significantly and the ophthalmoplegia OS had resolved; however, mild anterior segment inflammation remained. DISCUSSION Although ocular motor cranial neuropathy may occur following HZO, complete ophthalmoplegia is uncommon ( 1- 3). To our knowledge, only 16 cases ( including our own) have been previously reported in the English medical literature over the past 30 years ( 3- 9). ( Table 1.) The etiology of ocular motor cranial nerve involvement following HZO remains controversial. Third nerve involvement has been reported to occur with the greatest frequency, followed by sixth and fourth cranial nerve involvement ( 1,3). Edgerton suggested that ocular motor cranial nerve involvement arises from contiguous spread of inflammation from the trigeminal nerve to the cavernous sinus or superior orbital fissure ( 1). Carroll and Mastaglia suggested that the pattern of onset and rate of recovery is indicative of demyelinating disease ( 6). Lavin et al. ( 7) reported the presence of demyelination of the third, fourth, and fifth nerves in an autopsy case; several of the vessels supplying those nerves showed perivascular monocytic infiltrates ( 7). Garg et al. ( 9) thought that microinfarction of the cranial nerves was a possible etiology, based on pupil- sparing of the third nerve in their case ( 9). Naumann et al. ( 10) found chronic inflammatory cell infiltration in the long posterior ciliary vessels and nerves of 21 enucleated eyes affected by HZO, suggesting that the neuropathy arises from an occlusive vasculitis ( 10). We favor secondary inflammatory vascular disease as the etiology for zoster- related ophthalmoplegia. The prognosis for recovery after complete ophthalmoplegia following HZO is good. Of the 16 total reported cases ( including our three cases), nine cases had adequate documented ophthalmic follow- up. All of these nine cases showed significant improvement within 2 months and complete or near resolution within 18 months. The effect of steroid or antiviral treatment on HZO-associated ophthalmoplegia has not been formally studied. Of the 16 total cases of HZO- associated complete ophthalmoplegia reported in the literature, eight ( 50%) patients did not have documented treatment, one ( 6%) had definitely received no treatment, and seven ( 44%) TABLE 1. Review of 16 cases: Complete Ophthalmoplegia following Herpes Zoster Ophthalmicus ( HZO) Case no. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 Author, Year Chang- Godinich, 1997 Chang- Godinich, 1997 Chang- Godinich, 1997 Bourke, 1994 Archambault, 1993 Archambault, 1993 Archambault, 1993 Garg, 1992 Lavin, 1984 Carroll, 1979 Kattah, 1978 Kattah, 1978 Marsh, 1977 Marsh, 1977 Marsh, 1977 Marsh, 1977 Age ( yr) 82 72 75 63 73 63 70 Young 57 55 72 54 Unknown Unknown Unknown Unknown Sex F F M F F M M F F M F F Underlying condition Rheumatoid arthritis Chronic lymphocytic leukemia MI Healthy - - - - Atherosclerosis Pharyngitis - Hodgkin's lymphoma stage IIIB - - - - Time to onset" ( wk) 1 2 0.5 0.7 2 2 1.5 0.5 5 0.7 0.7 0.1 - - - - Proptosis No No Yes Yes - - - - - - Yes Yes Yes Yes Yes No Optic neuropathy No Yes No Yes - - - - - Yes Yes Yes - - - - Uveitis No No Yes Yes - - - - - - Yes No - .- - - Treatment Acyclovir, steroids Acyclovir, steroids Topical steroids Acyclovir, steroids - - - - Steroids, Cytoxan None Steroids Steroids - - - - Time to resolution ( mo) 1 > 2 2 1.5- 12 2-> 12 2-> 17 2- 3 - MI 1- 18 1 E - - - - E, evisceration; F, female; M, male; MI, myocardial infarction. " Onset following appearance of rash. J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 COMPLETE OPHTHALMOPLEGIA AFTER ZOSTER OPHTHALMICUS 265 received treatment with some form of steroid. Three of these seven ( 43%) patients were treated with steroid alone, three ( 43%) were treated with steroid in combination with acyclovir, and one ( 14%) was treated with steroid in combination with cyclophosphamide. Five of these seven cases ( 71%) showed marked improvement in ophthalmoplegia within 2- 3 months time; two of these five ( 29%) patients were maintained on steroids alone without addition of acyclovir. One of the steroid- treated patients suffered serious complications of microbial keratitis requiring evisceration, and one cyclophosphamide and steroid- treated patient suffered a myocardial infarction. The potential effect of antiviral treatment alone is unknown, as all three patients treated with antiviral therapy were also treated with steroids. HZO- associated complete ophthalmoplegia occurs mostly in individuals > 50 years of age ( range, 54- 82 years). Onset of ophthalmoplegia is usually within 1- 2 weeks following development of cutaneous HZO ( range, 0.1- 5 weeks). Of the cases with adequate documentation of ophthalmic findings, proptosis ( seven of 10 cases), optic neuropathy ( five of seven cases), and anterior uveitis ( three of six cases) were observed in addition to ophthalmoplegia. Ophthalmologists should be aware that complete ophthalmoplegia may follow cutaneous HZO and that the prognosis for recovery within several months is good. This work was supported in part by an Unrestricted Grant from Research to Prevent Blindness, Inc., New York, NY, U. S. A. and the Baylor Neuro- Ophthalmology Academic Fund. REFERENCES 1. Edgerton AE. Herpes zoster ophthalmicus: report of cases and review of literature. Arch Ophthalmol 1945; 34: 40- 62, and 114- 53. 2. Wanock LW, Liesegang TJ. Complications of herpes zoster opthalmicus. Arch Ophthalmol 1983; 101: 42- 5. 3. Marsh RJ, Dulley, B, Kelly, V. External ocular motor palsies in ophthalmic zoster: a review. Br J Ophthalmol 1977; 61: 677- 82. 4. Archambault P, Wise JS, Rosen J, Polomeno RC, Auger N. Herpes zoster ophthalmoplegia: report of six cases. J Clin Neuro- Ophthalmol 1993; 8: 185- 91. 5. Kattah JC, Kennerdell JS. Orbital apex syndrome secondary to herpes zoster ophthalmicus. Am J Ophthalmol 1978; 85: 378- 82. 6. Carroll WM, Mastaglia FL. Optic neuropathy and ophthalmoplegia in herpes zoster oticus. Neurology 1979; 29: 726- 9. 7. Lavin PJM, Younkin SG, Kori SH. The pathology of ophthalmoplegia in herpes zoster opthalmicus. Neuro- Ophthalmology 1984; 4: 75- 80. 8. Bourke RD, Pyle J. Herpes zoster opthalmicus and the orbital apex syndrome. Aust NZ J Ophthalmol 1994; 22: 77- 80. 9. Garg RK, Kar AM, Jain AK. Herpes zoster opthalmicus with complete external ophthalmoplegia. J Assoc Phys India 1992; 40: 486- 97. 10. Naumann G, Gass JDM, Font RL. Histopathology of herpes zoster ophthalmicus. Am J Ophthalmol 1968; 65: 221- 2 J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 |