| OCR Text |
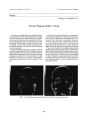
Show Journal of Neuro- Ophthalmology 17( 4): 254- 256, 1997. © 1997 Lippincott- Raven Publishers, Philadelphia A Case of Bilateral Optic Neuropathy and Recurrent Transverse Myelopathy Associated With Perinuclear Anti- Neutrophil Cytoplasmic Antibodies ( p- ANCA) Takayuki Harada, M. D.,* Tsutomu Ohashi, M. D.,* Chikako Harada, M. D.,* Kazuhiko Yoshida, M. D.,* Shiroh Maguchi, M. D., t Fumio Moriwaka, M. D.,$ and Hidehiko Matsuda, M. D.* A case involving a 52- year- old man having bilateral optic neuropathy and recurrent transverse myelopathy is reported. His clinical features resembled multiple sclerosis, but neuroimag-ing failed to show evidence of demyelination or inflammation in the brain or the optic nerves. The patient experienced sudden visual loss despite massive steroid therapy. Positive perinuclear anti- neutrophil cytoplasmic antibody ( p- ANCA) is suggestive that optic neuropathy and recurrent transverse myelopathy may have been caused by some common inflammatory processes associated with p- ANCA, however, having a different etiology from multiple sclerosis. Key words: Perinuclear anti-neutrophil cytoplasmic antibody ( p- ANCA)- Optic neuropathy- Transverse myelopathy- Multiple sclerosis. Anti- neutrophil cytoplasmic antibodies ( ANCAs) are divided into two subtypes differentiated by their staining patterns on indirect immunofluorescence ( 1). Cytoplasmic anti- neutrophil cytoplasmic antibodies ( c- ANCAs) have been described as sensitive and specific markers for active Wegener's granulomatosis ( 2), whereas perinuclear anti- neutrophil cytoplasmic antibodies ( p- ANCAs) occur in a wide range of diseases, including ulcerative colitis ( 3- 5), autoimmune liver disease ( 6,7), and rheumatoid arthritis ( 8- 11). However, ANCAs have received little attention in the ophthalmic literature ( 12), and reported cases of optic nerve involvement are rare ( 13). We present such a case. CASE REPORT In April 1995, a 52- year- old man noticed a weakness in both legs and had difficulty in walking, prompting a visit to the neurosurgery hospital. Magnetic resonance ( MR) imaging with gadolinium enhancement revealed Manuscript received October 8, 1996; accepted January 13, 1997. From the Departments of "" Ophthalmology, fOtolaryngology, and ^ Neurology, Hokkaido University School of Medicine, Sapporo, Japan. Address correspondence and reprint requests to Dr. Takayuki Harada, Department of Degenerative Neurological Diseases, National Institute of Neuroscience, NCNP, 4- 1- 1 Ogawahigashi- cho, Kodaira, Tokyo, 187 Japan. spinal cord swelling from the sixth to ninth thoracic vertebra on Tl- weighted image. A diagnosis of acute transverse myelopathy was made. After steroid therapy ( 100 mg of intravenous methylpredonisolone), the swelling resolved and the patient's functional ability returned to normal. On July 10, the patient experienced visual loss in his right eye and visited the local hospital. He had no history of retrobulbar pain or attacks of optic neuritis. His best-corrected visual acuity was 20/ 100 in the right eye; however, no abnormalities were found in the fundus. There was a reduced pupillary response to light in the right eye. Computed tomographic scans and MR imaging of the brain with gadolinium enhancement revealed no abnormalities. The patient was diagnosed as having retrobulbar optic neuritis associated with multiple sclerosis ( MS) and underwent steroid treatment ( 100 mg of intravenous methylpredonisolone). Since there was no visual recovery, he was referred to our clinic on August 8. Our initial examination revealed the best- corrected visual acuity to be hand motion in the right eye and 20/ 15 in the left eye. The eye movement, anterior segments, intraocular pressure, and fundus were normal. We could not carry out a visual field test of the right eye because of severe visual disturbance. The visual field of the left eye was found to be normal. There was a reduced pupillary response and a mild afferent pupillary defect in the right eye. Fluorescein angiography showed a late staining on the right disc; however, no other specific abnormalities were noticed ( Fig. 1). Laboratory studies were positive for antinuclear antibodies ( ANAs) ( 1: 40, normal range < 1; 20), anti- Ro ( SS- A) antibodies ( 1: 55, normal range < 1: 20), and anti- La ( SS- B) antibodies ( 1: 82, normal range < 1: 25); however, results for anti- DNA antibodies ( 1.6 IU/ mL, normal range < 4 IU/ mL), anti- RNP antibodies ( 1: 2.8, normal range < 1: 7), anti- Sm antibodies ( 1: 0.1, normal range < 1: 5), and anticardiolipin ( 32 GPI antibodies ( 3.3 U/ mL, normal range < 3.5 U/ mL) were not positive. At that point, examinations for Sjogren's syndrome were carried out ( 14). The result of Schirmer's test was 11 to 13 mm ( normal > 10 mm) in 5 minutes, and 254 OPTIC NEUROPATHY ASSOCIATED WITH p- ANCA 255 FIG. 1. Fluorescein angiography showing no abnormalities except a late staining of the right optic disc. the rose bengal test showed the absence of punctate keratoconjunctivitis in both eyes. In addition, a sialogram of the parotid gland showed normal findings, and other manifestations of connective tissue disease were absent. Steroid treatment ( 15) was started with 1000 mg of intravenous methylprednisolone daily for 3 days, followed by oral prednisolone for 21 days ( 60 mg for 4 days, 40 mg for 3 days, 30 mg for 4 days, 20 mg for 3 days, 10 mg for 4 days, and 5 mg for 3 days); however, no beneficial effect was seen. On August 25, the color of the optic disc was completely pale ( Fig. 2) and his visual acuity was zero in the right eye. On October 24, the patient complained of a decrease in visual acuity in the left eye. Examination revealed the best- corrected visual acuity to be no light perception in the right eye and 20/ 100 in the left eye. Panel D- 15 test of the left eye revealed deutanomaly. The visual field testing of the left eye demonstrated central scotoma ( Fig. 3). Visual evoked response testing revealed decreased amplitude in the left eye. Steroid therapy was tried again as described earlier ( 1000 mg of intravenous methylprednisolone daily for 3 days, and followed by oral prednis- FIG. 2. Fundus photograph of the right eye showing optic atrophy. olone for 21 days), but the left eye progressed to permanent visual deficit, and both optic discs were completely pale on November 25. In January 1996, he again had difficulty in walking. A spinal tap revealed increased mononuclear cells ( 10/ mm, normal range < 5/ mm), elevated concentration of protein ( 0.116 g/ L, normal range < 0.045 g/ L), and IgG ( 15.2 mg/ dL, normal range < 4 mg/ dL). In addition, results for the serum p- ANCA were positive ( 1: 8, normal range < 1: 1), whereas results for c- ANCA were negative (< 1: 1) ( 16). MR imaging showed spinal cord swelling from the sixth to ninth thoracic vertebra. Despite the steroid therapy just described, functional disorders remained unchanged. DISCUSSION We believe this case to be the first in the ophthalmic literature, presenting bilateral optic neuropathy resulting in a complete visual loss, having both MS- like symptoms and p- ANCA. Optic neuritis treatment trial ( ONTT) of 448 patients ( 17) concluded that in a typical patient presenting with sudden visual loss with progression of symptoms of 1 week or less accompanied by pain with either a swollen or normal optic disc, it is unlikely that any ancillary studies will define an etiologic factor for optic neuritis other than demyelination. In addition, they reported that MR imaging showed changes consistent with demyelination of the brain in 48.7% of the patients. Contrary to a typical patient, the patient described here experienced a complete loss of vision without remission despite the massive steroid treatments given. Additionally, neuroim-aging failed to show lesions consistent with MS plaques in the brain or optic nerves ( 18). Certainly, the current case fulfilled the clinical criteria for a diagnosis of MS ( 19) since there were four attacks involving two different parts of the central nervous system, separated by a period of 3 months. However, based on these findings, the pathogenesis of optic nerve involvement seems to have been ischemic necrosis secondary to small vessel vasculitis of the optic nerves, rather than the reversible edema- J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 256 T. HARADAETAL. tous changes caused by demyelinating processes. Laboratory studies were positive for ANAs, SS- A antibodies, SS- B antibodies, and p- ANCA. In the ONTT, about 16% of patients were positive for ANAs; however, this finding had no prognostic significance when ANA- positive patients were compared with ANA- negative patients ( 17). In addition, there were no other manifestations of connective tissue disease, including Sjogren's syndrome, in this case ( 14). Thus, the presence of p- ANCA seems to be a key to understanding the pathogenesis of this disorder. This assumption also concurs with the previous reports that p- ANCA may have a pathogenic role in necrotizing systemic vasculitis ( 20,21). In addition, we recently found a significantly higher p- ANCA- positive rate ( 46.2%) among patients with a diagnosis of optic-spinal form of MS ( OpS- MS) compared with that ( 0%) among the 26 patients with conventional MS ( 22). Thus, together with our previous studies, ( 18,23) this case also suggests the possibility that optic neuropathy and recurrent transverse myelopathy in some of OpS- MS patients may be caused by some common inflammatory processes associated with p- ANCA and other autoantibodies rather than from MS. Finally, care must be taken when making a diagnosis of optic neuropathy because the visual prognosis and treatment are different according to the etiology. Although assessment of their diagnostic value has to wait further characterization, it may be necessary in the future to determine whether the patient has evidence of p- ANCA as well as phospholipid- directed autoantibodies before making a diagnosis of MS. REFERENCES 1. Kallenberg CGM, Mulder AHL, Tervaert JWC. Antineutrophil cytoplasmic antibodies: a still- growing class of autoantibodies in inflammatory disorders. Am J Med 1992; 93: 675- 82. 2. Van der Woude FJ, Rasmessen N, Labatto S, et al. Autoantibodies to neutrophils and monocytes: a new tool for diagnosis and a marker of disease activity in Wegener's granulomatosis. Lancet 1985; ii: 425- 9. 3. Saxon A, Sharahan F, Landers C, et al. A distinct subset of antineutrophil cytoplasmic antibodies is associated with inflammatory bowel disease. J Allergy Clin Immunol 1990; 86: 202- 10. 4. Rump JA, Scholmerich J, Gross V, et al. A new type of perinuclear antineutrophil cytoplasmic antibody ( p- ANCA) in active ulcerative colitis but not in Crohn's disease. Immunology 1991; 181: 406- 13. 5. Duerr RH, Targan SR, Landers CJ, et al. Antineutrophil cytoplasmic antibodies in ulcerative colitis: comparison with other colitis/ diarrheal illnesses. Gastroenterology 1991; 100: 1590- 6. 6. Duerr RH, Targan SR, Landers CJ, et al. Neutrophil cytoplasmic antibodies: a link between primary sclerosing cholangitis and ulcerative colitis. Gastroenterology 1991; 100: 1385- 91. 7. Cohen Tervaert JW, Mulder AHL, Horst G, et al. Antineutrophil cytoplasmic antibodies in autoimmune liver disease. Gastroenterology 1992; 102: 1090. 8. Mulder AHL, Horst G, van Leeuwen MA, et al. Granulocyte-specific antinuclear antibodies are directed against a granular compound of the granulocyte. Clin Rheumatol 1990; 9: 578. 9. Christenson VD, Dooley MA, Allien NB. Discrimination of antineutrophil antibodies from antinuclear antibodies using immunofluorescence on neutrophils and HL60 cells. J Rheumatol 1991; 18: 575- 9. 10. Lassoued S, Sixou L, Oksman F, et al. Antineutrophil cytoplasmic antibodies and antibodies to myeloperoxidase in rheumatoid arthritis. Arthritis Rheum 1991; 34: 1069- 70. 11. Savige JA, Gallicchio MC, Stockman A, et al. Antineutrophil cytoplasmic antibodies in rheumatoid arthritis. Clin Exp Immunol 1991; 86: 92- 8. 12. Soukiasian SH, Foster CS, Niles JL, et al. Diagnostic value of antineutrophil cytoplasmic antibodies in scleritis associated with Wegener's granulomatosis. Ophthalmology 1992; 99: 125- 32. 13. Belden CJ, Hamed LM, Mancuso AA. Bilateral isolated retrobulbar optic neuropathy in limited Wegener's granulomatosis. J Clin neuroophthalmol 1993; 13: 119- 23. 14. Harada T, Ohashi T, Miyagishi R, et al. Optic neuropathy and acute transverse myelopathy in primary Sjogren's syndrome. Jpn J Ophthalmol 1995; 39: 162- 5. 15. Beck RW, Cleary PA, Anderson MM, et al. A randomized, controlled trial of corticosteroids in the treatment of acute optic neuritis. N Engl J Med 1992; 326: 581- 8. 16. Maguchi S, Takizawa M, Ryu T, et al. Clinical significance of antineutrophil antibody ( ANCA) for the diagnosis of Wegener's granulomatosis in the early stage. AmJRhinol 1994;( suppl) 9; 369. 17. Optic Neuritis Study Group. The clinical profile of optic neuritis: experience of the Optic Neuritis Treatment Trial. Arch Ophthalmol 1991; 109: 1673- 8. 18. Fukazawa T, Miyasaka K, Tashiro K, et al. MRI findings of multiple sclerosis with acute transverse myelopathy. J Neurol Sci 1992; 110: 27- 31. 19. Poser CM, Paty DW, Scheinberg L, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol 1983; 13: 227- 31 20. Chalk CH, Homburger HA, Dyck PJ. Anti- neutrophil cytoplasmic antibodies in vasculitic peripheral neuropathy. Neurology 1993; 43: 1826- 7. 21. Falk RJ, Terrell RS, Charles LA, et al. Anti- neutrophil cytoplasmic antibodies induce neutrophils to degranulate and produce oxygen radicals in vitro, Proc Natl Acad Sci USA 1990; 87: 4115- 9. 22. Fukazawa T, Hamada T, Kikuchi S, et al. Antineutrophil cytoplasmic antibodies and the optic- spinal form of multiple sclerosis in Japan. J Neurol Neurosurg Psychiatry 1996; 61: 203- 4. 23. Harada T, Ohashi T, Fukazawa T, et al. Visual function in patients with optic neuritis associated with acute transverse myelopathy in multiple sclerosis. Jpn J Ophthalmol 1995; 39: 290- 4. J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 |