| OCR Text |
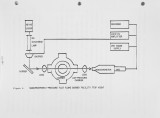
Show ; COMBUSTION/INHIBITION PHYSICO-CHEMISTRY OF COAL-DERIVED SYNGAS/AIR FLAMES R. D. Daley, J. A. Withum, J. J. Warchol, and J. J. Reuther Fuels and Combustion Laboratory The Pennsylvania State University University Park, PA 16802 ABSTRACT The first results of a comprehensive and systematic experimental program on low-Btu coal-derived syngas/air combustion, underway at The Pennsylvania State University Fuels and Combustion Laboratory, are reported. Gaseous fuels [CH,, CO, H2, NH ] and diluents [CO , N , H O ] were parametrically mixed together, simulating the compositions of near-commercial, coal-derived syngases. The physical [temperature] and chemical [transient and stable species concentrations] microstructure profiles of over 50 non-adiabatic flat Lurgi- and Wellman-Galusha-air flames were determined. From an applied-combustion research viewpoint, the most significant findings were that: (a) the fuel-bound-nitrogen (FBN=NH«) conversion efficiency, and, therefore, the concentration of NO in the exhaust, maximized at a stoichiometry (equivalence ratio) of 1.2 for both syngas/air flames, and (b) at the same FBN doping-level, more NO was produced in Lurgi/air flames than in Wellman-Galusha/air flames. These findings may have an important bearing on: (a) staged-combus-tion- for-NOX-control-strategies which are based on results from combustion research on flames fueled with individual syngas components that showed the FBN conversion efficiency peaking at stoichiometries of *k 1.0, and (b) coal-derived syngas fuel, and therefore, coal-gasifier, selection, respectively. tJL-1- |