| OCR Text |
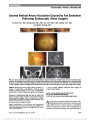
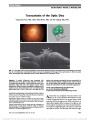
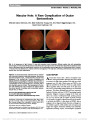
Show Toxocariasis of the Optic Disc Yong Joon Kim, MD, Chan Hee Moon, MD, Jee Ho Chang, MD, PhD Abstract: A healthy 46-year-old man presented with decreased vision in the right eye after ingestion of raw meat. On funduscopic examination, a cystic lesion was found on an edematous right optic disc with adjacent serous retinal detachment. Optical coherence tomography confirmed a peri-papillary serous retinal detachment and a well-demarcated cystic lesion (200 · 200 · 500 mm) in the right eye. The patient had moderate eosinophilia and was seropositive for anti-Toxocara IgG antibody. Diagnosed with ocular toxocaria-sis, he was treated with systemic corticosteroids and alben-dazole with improvement in vision and fundus appearance. Journal of Neuro-Ophthalmology 2013;33:151-152 doi: 10.1097/WNO.0b013e3182893ff9 © 2013 by North American Neuro-Ophthalmology Society A46-year-old man complained of decreased vision in his right eye for 3 days associated with headache. He had a history of eating raw beef liver over the past month. Visual acuity was 20/40 in the right eye and 20/20 in the left eye. A relative afferent papillary defect was present in his right eye, and anterior segment examination was normal. Automated perime-try revealed an enlarged blind spot in the right eye, and the left visual field was normal. The left fundus was normal; the right FIG. 1. A. The right fundus shows peripapillary serous retinal detachment and a small cystic lesion (arrowheads) on the optic disc. B. A 3-dimensional reconstructed optical coherence tomography (OCT) image of the cystic lesion. C. Both the cystic lesion (arrowheads) and peripapillary serous retinal detachment are visualized on OCT. Department of Ophthalmology, Soonchunhyang University College of Medicine, Bucheon Hospital (YJK, CHM, JHC), Bucheon, South Korea. The authors report no conflicts of interest. Supplemental digital content is available for this article. Direct URL citations appear in the full text and are provided in the HTML and PDF versions of this article on the journal's Web site (www. jneuro-ophthalmology.com). Address correspondence to Jee Ho Chang, MD, PhD, Department of Ophthalmology, Soonchunhyang University College of Medicine, Bucheon Hospital, #1174 Jung-Dong, Wonmi-Gu, Bucheon 420-767, South Korea, E-mail:jhchang@schmc.ac.kr Kim et al: J Neuro-Ophthalmol 2013; 33: 151-152 151 Photo Essay Section Editor: Timothy J. McCulley, MD Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. fundus showed optic disc edemawith a cystic lesion at its center and adjacent serous retinal detachment (Fig. 1A). Spectral-domain (Cirrus HD-OCT, Carl Zeiss Meditec Inc., Dublin, CA) optical coherence tomography (OCT) revealed a peripapil-lary serous retinal detachment and a well-demarcated cystic lesion (200 · 200 · 500 mm) overlying the right optic disc (Figs 1B and 1C; see Videos 1 and 2, Supplemental Digital Contents 1 and 2, http://links.lww.com/WNO/A62 and http://links.lww.com/WNO/A63). Complete blood count showed moderate eosinophilia of 1,561 cells/mL (normal ,350 cells/mL). Liver function tests, C-reactive protein, and reactive plasma reagin were within normal limits. Rheumatoid factor was positive, but antinu-clear antibody was negative as were antibody tests for herpes simplex, varicella zoster, cytomegalovirus, and human immu-nodeficiency virus. Stool examinations were negative for helminthic and protozoan ova and larvae. Antibody tests for Toxoplasma, Taenia solium, Sparganum, Clonorchis sinen-sis, and Paragonimus westermani were negative, but positive for anti-Toxocara IgG using an enzyme-linked immunosor-bent assay (ELISA). The patient was diagnosed with ocular toxocariasis and treated with systemic corticosteroids and albendazole. One week later, visual acuity was 20/25 in the right eye. OCT showed the cystic lesion to be resolving, and there was less optic disc edema and peripapillary subretinal serous fluid (Fig. 2). Toxocariasis results from a parasitic infection by the ascarid larvae of Toxocara canis or Toxocara cati, which are usually found in the intestinal tract of dogs and cats (1). The adult nematode produces eggs, which are released into the environ-ment via the host's stool. The main source of eggs is young dogs, which become infected mainly through transplacental or transmammary transmission (2). Human infection is caused by accidental ingestion of embryonated eggs, via consumption of contaminated soil, raw vegetables, or meat (3). After ingestion, the embryonated eggs hatch in the duodenum and release larvae that penetrate the intestinal mucosal epithelium. They are carried to a wide variety of tissues, including the liver, lungs, muscles, brain, heart, and eyes. In human tissue, through the circulation, the larvae remain developmentally arrested, show-ing no growth or morphological differentiation and cannot complete their life cycle (4). Clinical manifestations of toxocariasis are dependent on the parasite load, the immune response, and the migration pattern of the Toxocara larva (5). Ocular larva migrans is caused by the migration of Toxocara larvae through retinal and choroidal blood vessels into the posterior segment of the eye (6). Ocular toxocariasis usually presents as peripheral granuloma, posterior granuloma, or nematode endophthalmitis (7). Disease-related complications of ocular toxocariasis include amblyopia, cata-ract, retinal folds and detachment, macular scarring, and phthi-sis bulbi (1). Serological testing, using an ELISA, is currently the standard technique for diagnosing ocular toxocariasis (8). Unlike viscera larva migrans, which shows marked periph-eral blood eosinophilia, in some ocular larva migrans patients, the number of eosinophils may be normal or mildly elevated even in patients with severe ocular manifes-tations. This is likely due to the blood-ocular barrier (9). There are few reports of OCT findings in ocular toxocariasis (10), but to the best of our knowledge, Toxocara overlying the optic nerve previously has not been reported. REFERENCES 1. Woodhall D, Starr MC, Montgomery SP, Jones JL, Lum F, Read RW, Moorthy RS. Ocular toxocariasis: epidemiologic, anatomic, and therapeutic variations based on a survey of ophthalmic subspecialists. Ophthalmology. 2012;119:1211-1217. 2. Wisniewska-Liqier M, Wozniakowska-Gesicka T, Sobolewska- Dryjanska J, Markiewicz-Jozwiak A, Wieczorek M. Analysis of the course and treatment of toxocariasis in children-a long-term observation. Parasitol Res. 2012;110:2363-2371. 3. Kwon SI, Lee JP, Park SP, Lee EK, Huh S, Park IW. Ocular toxocariasis in Korea. Jpn J Ophthalmol. 2011;55:143-147. 4. Maizels RM, Tetteh KK, Loukas A. Toxocara canis: genes expressed by the arrested infective larval stage of parasitic nematode. Int J Parasitol. 2000;30:495-508. 5. Glickman LT, Schantz PM. Epidemiology and pathogenesis of zoonotic toxocariasis. Epidemiol Rev. 1981;3:230-250. 6. Taylor MR. The epidemiology of ocular toxocariasis. J Helminthol. 2001;75:109-118. 7. Pivetti-Pezzi P. Ocular toxocariasis. Int J Med Sci. 2009;6:129-130. 8. Magnaval JF, Glickman LT, Dorchies P, Morassin B. Highlights of human toxocariasis. Korean J Parasitol. 2001;39:1-11. 9. Watthanakulpanich D. Diagnostic treads of human toxocariasis. J Trop Med Parasitol. 2010;33:44-52. 10. Higashide T, Akao N, Shirao E, Shirao Y. Optical coherence tomographic and angiographic findings of a case with subretinal toxocara granuloma. Am J Ophthalmol. 2003;136:188-190. FIG. 2. One week after treatment, optical coherence tomography demonstrates that the lesion overlying the optic nerve is resolving with decreased optic disc edema and peripapillary serous subretinal fluid. 152 Kim et al: J Neuro-Ophthalmol 2013; 33: 151-152 Photo Essay Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |