| OCR Text |
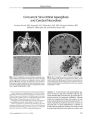
Show LETTERS TO THE EDITOR Increased Intracranial Pressure Associated With Ophthalmoplegia, Ataxia, and Areflexia To the Editor: Patients with idiopathic intracranial hypertension ( IIH) typically present with normal examination findings except for papilledema and sixth nerve palsy. This report describes a patient with external ophthalmoplegia, ataxia, areflexia, and mild proximal weakness in addition to the other findings of IIH. A 29- year- old woman was admitted with headache and visual loss three weeks after an abortion. The pregnancy was uncomplicated until the end of the fifth month, when she was admitted to a hospital with the diagnosis of abruptio placenta. She was discharged one week later without any symptoms. Two weeks later, she was referred to our institution with headache, neck pain, and blurred vision. We found visual acuities of 20/ 50 OD and finger counting OS. Optic disc edema with peripapillary hemorrhages was present OU. There was a complete limitation of eye movements in all directions. No ptosis was evident and the pupils were reactive. She had gait ataxia and mild proximal weakness ( Medical Research Council grade 4) in the upper and lower extremities. Deep tendon reflexes were absent. The rest of the neurologic examination was normal. Routine laboratory tests, cranial computed tomography, magnetic resonance imaging ( MRI), magnetic resonance venography, and cerebral angiography were normal. Distension of the perioptic nerve sheaths was detected in the orbital MRI. Lumbar puncture ( LP) revealed an opening pressure of 350 mmH20 with normal protein, glucose, and cell count. Motor and sensory nerve conduction studies ( baseline and repeated two weeks later) were normal. No antibodies against Campylobacter jejuni or Gqlb were detected in the serum or cerebrospinal fluid ( CSF). Because the ophthalmoplegia rapidly improved within 24 hours of the LP, a second LP was performed on the second day of admission, which showed an opening pressure of 300 mm H20. After drainage of 40 mL of CSF, the patient was begun on treatment with acetazolamide ( initially 250 mg twice daily, increased gradually to four times daily) and methylprednisolone ( 1 mg/ kg per day). On the fifth day after admission, the motor examination, gait, and eye movements were completely normal. Headache was relieved. Despite the recovery of these symptoms, the opening pressure of a third LP performed on day 7 of admission was still elevated at 270 mm H20. Because no improvement was detected in visual acuity, optic nerve fenestration was performed on the right eye. Corticosteroid treatment was discontinued and the patient was discharged on acetazolamide treatment. Examination three months later showed a visual acuity of 20/ 30 OD and 20/ 200 OS, with bilateral optic nerve pallor more prominent OS. The rest of the neurologic examination was normal. There are numerous reports of tinnitus, neck stiffness, paresthesias, ataxia, and third, fourth, fifth, and seventh cranial nerve palsies in IIH ( 1- 5). Kidron et al ( 6) used the term " malignant pseudotumor cerebri" for their two patients having rapidly progressive visual loss, ophthalmoplegia, and areflexia. Friedman et al ( 7) reported nine cases of IIH with external ophthalmoplegia, including four cases from the former literature ( 6,8). In common with our patient, three have had a miscarriage. Ophthalmoplegia, ataxia, and areflexia improved rapidly, but significant visual loss remained in five of the patients. The explanation for the ophthalmoplegia in these IIH cases remains unknown, but increased CSF pressure itself may cause it ( 7). Ataxia may be the result of a relative orthostatic hypotension ( 1). We have no explanation for the presence of weakness and areflexia. Fisher syndrome could have been the diagnosis in this patient, but the presence of normal CSF formula, normal electrophysiologic findings, and negative antibody results make this diagnosis highly unlikely. We acknowledge the many reports of Guillain- Barre syndrome or Fisher syndrome with papilledema and elevated CSF pressure ( 9,10), and that neither the CSF nor electrophysiologic findings by themselves are enough to rule out these diagnoses, but it would be unusual to have such rapid recovery. Ethem Murat Arsava, MD Bedile Irem Cikrikci, MD Tulay Kansu, MD Department of Neurology Hacettepe University Faculty of Medicine Ankara, Turkey arsavaem@ hotmail. com REFERENCES 1. Round R, Keane JR. The minor symptoms of increased intracranial pressure: 101 patients with benign intracranial hypertension. Neurology 1988; 38: 1461- 1464. 2. Davenport RJ, Will RG, Galloway PJ. Isolated intracranial hypertension presenting with trigeminal neuropathy. J Neurol Neurosurg Psychiatry 1994; 57: 381. 3. Selky AK, Dobyns WB, Yee RD. Idiopathic intracranial hypertension and facial diplegia. Neurology 1994; 44: 357. 4. Speer C, Pearlman J, Phillips PH, et al. Fourth cranial nerve palsy in pediatric patients with pseudotumor cerebri. Am J Ophthalmol 1999; 127: 236- 7. J Neuro- Ophthalmol, Vol. 24, No. 2, 2004 185 JNeuro- Ophthalmol, Vol. 24, No. 2, 2004 Letters to the Editor 5. McCammon A, Kaufman HH, Sears ES. Transient oculomotor paralysis in pseudotumor cerebri. Neurology 1981 ; 31: 182- 4. 6. Kidron D, Pomeranz S. Malignant pseudotumor cerebri. Report of two cases. JNeurosurg 1989; 71: 443- 5. 7. Friedman DI, Forman S, Levi L, et al. Unusual ocular motility disturbances with increased intracranial pressure. Neurology 1998; 50: 1893- 6. 8. Snyder DA, Frenkel M. An unusual presentation of pseudotumor cerebri. Ann Ophthalmol 1979; 11: 1823- 7. 9. Macaya A, Roig M, Fernandez JM, et al. Pseudotumor cerebri, spinal and radicular pain and hyporeflexia: a clinical variant of the Guillain- Barre syndrome? Pediatr Neurol 1988; 4: 120- 1. 10. Peterman AF, Daly DD, Dion FR. Infectious neuronitis ( Guillain- Barre Syndrome) in children. Neurology 1959; 9: 533- 9. Cat Scratch Disease in Two Brothers To the Editor: A nine- year- old boy noted decreased vision OD three days before examination. He had a flu- like illness two weeks earlier, which included intermittent fever and headaches. The family had recently acquired a four- month- old kitten, but no one was aware of being scratched. Visual acuity was 20/ 40 OD and 20/ 20 OS with a small right relative afferent pupillary defect. Automated perimetry revealed an enlarged blind spot OD and was normal OS. There were cells in the vitreous, more marked in the OS. Optic disc edema was present OD and there were foci of retinochoroiditis in both eyes ( Fig. 1). The erythrocyte sedimentation rate was 41 mm/ h and serum IgG for Bartonella henselae was positive, more than 1: 256. He was treated with 250 mg azithromycin per day for three weeks. His eight- year- old brother, who was not present during the initial examination and did not report visual loss, also had intermittent fever and headaches concurrent with his brother's symptoms. His serum IgM fori?, henselae was FIG. 1. Fundus photographs of the nine- year- old patient reveals optic disc swelling OD and areas of retinochoroiditis OS ( arrows). 186 FIG. 2. Fundus photograph of the nine- year- old's visually asymptomatic eight- year- old brother, who had a history of fever, headaches, and elevated Bartonella henselae titers, reveals mild opacification of the retinal nerve fiber layer and resolving parapapillary exudate ( arrows). 1: 512 with an IgG more than 1: 1024. He was asked to undergo an ophthalmic examination three weeks later. Visual acuity was 20/ 20 OU, with normal pupils. Mild vitritis was noted OU. Funduscopy revealed mild opacification of the retinal nerve fiber layer OD with resolving parapapillary exudates ( Fig. 2). The boy's mother and father had no history of visual loss, fevers, or headaches. Cat scratch disease has been increasingly noted as a cause of optic disc edema, retinochoroiditis, and neurore-tinitis ( 1,2). Coinfection of members in the same household would seem to be a likely result of exposure to an infected cat. However, concurrent ocular involvement has been rarely reported in members of the same family ( 3). Children and young adults appear to be at increased risk for systemic infection after exposure ( 4). Although at least one comparative trial studying lymph node volume has suggested the benefit of treatment with systemic antibiotics ( 5), the treatment of ocular manifestations of cat scratch disease remains controversial ( 4). Most patients will have visual improvement with conservative management ( 6), but irreversible visual loss has been noted ( 7). Members of the same household of a patient with cat scratch disease should be questioned for symptoms of systemic infection. In those with systemic symptoms, B. henselae titers should be obtained and ocular examination considered, even in those who are visually asymptomatic, particularly children, who may be less likely to report visual dysfunction. Rod Foroozan, MD Cullen Eye Institute Baylor College of Medicine Houston, Texas foroozan@ bcm. tmc. edu © 2004 Lippincott Williams & Wilkins Letters to the Editor JNeuro- Ophthalmol, Vol. 24, No. 2, 2004 REFERENCES Solley WA, Martin DF, Newman NJ, et al. Cat scratch disease: posterior segment manifestations. Ophthalmology 1999; 106: 1546- 53. Wade NK, Levi L, Jones MR, et al. Optic disk edema associated with peripapillary serous retinal detachment: an early sign of systemic Bartonella henselae infection. Am J Ophthalmol 2000; 130: 327- 34. Wong MT, Dolan MJ, Lattuada CP, Jr., et al. Neuroretinitis, aseptic meningitis, and lymphadenitis associated with Bartonella ( Rochali-maea) henselae infection in immunocompetent patients and patients infected with human immunodeficiency virus type 1. Clin Infect Dis 1995; 21: 352- 60. Cunningham ET, Koehler JE. Ocular bartonellosis. Am J Ophthalmol 2000; 130: 340- 9. Bass JW, Freitas BC, Freitas AD, et al. Prospective randomized double blind placebo- controlled evaluation of azithromycin for treatment of cat- scratch disease. Pediatr Infect Dis J 1998; 17: 447- 52. Rosen BS, Barry CJ, Nicoll AM, et al. Conservative management of documented neuroretinitis in cat scratch disease associated with Bartonella henselae infection. Aust N Z J Ophthalmol 1999; 27: 153- 6. Gray AV, Michels KS, Lauer AK, et al. Bartonella henselae infection associated with neuroretinitis, central retinal artery and vein occlusion, neovascular glaucoma, and severe vision loss. Am JOphthalmol 2004; 137: 187- 9. [CLcataractsurgery] Contralateral Amaurosis After Retrobulbar Anesthetic Injection To the Editor: I ( REW) recently performed cataract surgery on an 84- year- old woman who experienced transient contralateral visual loss after a retrobulbar block, presumably from ipsilateral optic nerve sheath penetration. Preoperatively, visual acuity was best- corrected to 20/ 60 OD, 20/ 30 OS, entirely attributable to cataract. After intravenous sedation, 3.5 mL of 2% lidocaine was injected into the right retrobulbar space. Fifteen minutes later, before commencing the surgery, the patient stated that she could not see. This was interpreted as visual loss on the injected side, and the patient was reassured. On completion of the case 25 minutes later ( 40 min after the retrobulbar injection), it became apparent that the patient could not see in either eye. Visual acuity was hand movements in the operated eye and no light perception in the unoperated eye. Motility was very limited in the operated eye and was normal except for a marked deficit of adduction in the unoperated eye. The pupil in the operated eye measured 6 mm in dim light and was minimally reactive to direct light; the pupil in the unoperated eye measured 7 mm and was unreactive to direct light. Indirect ophthalmoscopy of both eyes was normal. No intervention or treatment occurred. Over the next two hours, the visual acuity in the unoperated OS gradually recovered to its 20/ 30 baseline and the visual acuity in the operated OD ultimately improved to 20/ 25. Contralateral transient visual loss after retrobulbar anesthetic injection is a rare but well- recognized complication ( 1- 6) ( Table 1). Antoszyk and Buckley ( 3) reported the largest case series: three patients. In all three, there was evidence of contralateral visual loss and contralateral third nerve palsy, similar to the present case. They theorized that the anesthetic was injected into the subdural space of the optic nerve sheath and tracked along the ipsilateral optic nerve sheath to the orbital apex. As the anesthetic tracked posteriorly within the subdural space to the area of the chiasm, it interfered with conduction within the contralateral optic nerve and third cranial nerve. Brod ( 4) reported a case of ipsilateral central retinal artery occlusion and contralateral transient visual loss after a retrobulbar anesthetic injection in which computed tomography performed 90 minutes after the injection demonstrated an air bubble within the ipsilateral optic nerve sheath, consistent with intra- nerve sheath injection. The proposed mechanism of intra- optic nerve sheath injection and subsequent tracking of anesthetic into the intracranial subdural space is consistent with that observed in contrast orbitography ( 7- 10), a technique used in the pre-modern imaging era to delineate orbital anatomy and tu- TABLE 1. Contralateral transient visual loss after retrobulbar anesthetic injection Authors Patient age/ gender 67/ M 68/ M 59/ M 58/ M 64/ F 57/ M 68/ F 74/ M 63/ M 84/ F Procedure Keratoplasty resuture OS Penetrating keratoplasty OD Retinal reattachment OD Cataract extraction OS Cataract extraction OD Cataract extraction OD Pars plana vitrectomy OS Cataract extraction OS Cataract extraction OS Cataract extraction OS Follette andLoCascio, 1985 ( 1) Antoszyk and Buckley, 1986 ( 2) Friedberg and Kline, 1986 ( 3) Brod, 1989( 4) Bisharaetal, 1990( 5) Lau et al, 2003 ( 6) Warwar et al, ( present letter) 187 JNeuro- Ophthalmol, Vol. 24, No. 2, 2004 Letters to the Editor mors. Local anesthetic and contrast material are injected into the retrobulbar space and plain film x- rays are taken. Reed ( 7) reported a patient with temporary vertical nystagmus and contralateral sixth cranial nerve palsy, and x- rays confirmed the presence of radiopaque material within the optic nerve sheath and the intracranial subdural space. Kaufer and Augustin ( 8) reported the presence of radiopaque material along the optic nerve and within the subdural space of the midbrain in a patient who remained asymptomatic during a three- hour observation period. Lombardi ( 9) reported contrast material in the intracranial subdural space in 3 ( 2%) of 150 orbitographies. All three patients experienced pain, nausea, and vomiting but no focal neurologic deficits. Although a 2% incidence of subdural penetration with retrobulbar injections in contrast orbitography may seem high relative to the number reported for retrobulbar anesthetic injections, orbitography allows visualization of the injected material; with retrobulbar anesthetic injections, nerve sheath injections and subdural penetrations may be asymptomatic and go unrecognized. Cardiovascular instability and apnea after retrobulbar blocks have been attributed to " brain stem anesthesia" ( 11- 13). In these cases, the anesthetic is thought to enter the cerebrospinal fluid through the subarachnoid space of the optic nerve sheath. Why some injections enter the subdural and others the subarachnoid space is unknown. Perhaps the depth of needle penetration, the force and volume of injection, or individual anatomic variations are explanations. We know of no reports of permanent contralateral visual loss or systemic neurologic sequelae after intra- optic nerve sheath injections of anesthetic agents. With the growing popularity of topical ocular anesthesia, complications related to retrobulbar injections will likely decrease. However, retrobulbar alcohol injections for blind, painful eyes are still used. Although we are not aware of any reports of intra- optic nerve sheath injection of alcohol, the effect on the contralateral nerve and/ or the central nervous system could be devastating. Ronald E. Warwar, MD Department of Surgery Wright State University School of Medicine Dayton, Ohio rwarwar@ warwareyegroup . com Eric K. Romriell, DO Department of Ophthalmology Grandview Medical Center Dayton, Ohio Eric A. Pennock, MD Department of Surgery Wright State University School of Medicine Dayton, Ohio REFERENCES 1. Follette JW, LoCascio JA. Bilateral amaurosis following unilateral retrobulbar block ( letter). Anesthesiology 1985; 63: 237- 8. 2. Antoszyk AN, Buckley EG. Contralateral decreased visual acuity and extraocular muscle palsies following retrobulbar anesthesia. Ophthalmology 1986; 93: 462- 5. 3. Friedberg HL, Kline, Jr. OR. Contralateral amaurosis after retrobulbar injection. Am J Ophthalmol 1986; 101: 688- 90. 4. Brod RD. Transient central retinal artery occlusion and contralateral amaurosis after retrobulbar anesthetic injection. Ophthal Surg 1989; 20: 643- 6. 5. Bishara SA, Estrin I, Rand WJ. Immediate contralateral amaurosis after retrobulbar anesthesia. Ann Ophthalmol 1990; 22: 63- 5. 6. Lau LI, Lin PK, Hsu WM, et al. Ipsilateral globe penetration and transient contralateral amaurosis following retrobulbar anesthesia. Am J Ophthalmol 2003; 135: 251- 2. 7. Reed JW, MacMillan, Jr. AS, Lazenby GW. Transient Neurologic complication of positive contrast orbitography. Arch Ophthal 1969; 81: 508- 11. 8. Kaufer G, Augustin G. Orbitography. Report of a complication with use of water- soluble contrast material. Am J Ophthalmol 1966; 61: 795- 8. 9. Lombardi G. Radiology in neuro- ophthalmology. Baltimore: Wil-liams & Wilkins Co., 1967: 6- 8. 10. Drysdale DB. Experimental subdural retrobulbar injection of anesthesia. Ann Ophthalmol 1984; 16: 716- 8. 11. Hamilton RC. Brain stem anesthesia following retrobulbar blockade. Anesthesiology 1985; 63: 688- 90. 12. Chang JL, Gonzalez- Abola E, Larson CE, et al. Brain stem anesthesia following retrobulbar block ( letter). Anesthesiology 1984; 61: 789- 90. 13. Meyers EF, Ramirez RC, Boniuk I. Grand mal seizures after retrobulbar block. Arch Ophthalmol 1978; 96: 847. Eye Movement Abnormalities in a Case of X- Linked Dystonia- Parkinsonism ( Lubag) To the Editor: X- Linked dystonia- Parkinsonism is a rare movement disorder first described in 1976 on the island of Panay in the Philippines ( 1). Dubbed by local inhabitants as " lubag" for its characteristic twisting movements, it afflicts males in the third to fifth decades ( 2,3). We describe a man afflicted with lubag who displays eye movement abnormalities that have not been previously reported. A 54- year- old man from the island of Panay, province of Capiz, the Philippines, had writing difficulties secondary to dystonic hand posturing. This was followed by slowly progressive speech difficulties, dystonic limb and trunk movements, cogwheel rigidity, and postural instability ( 4). When first examined in 1989, he had no ophthalmic symptoms, and ophthalmologic examination then was remarkable only for horizontal saccadic pursuit movements. Over the next seven years, he was treated with 25 mg carbidopa/ 250 mg levodopa three times daily, 100 mg 188 © 2004 Lippincott Williams & Wilkins Letters to the Editor JNeuro- Ophthalmol, Vol. 24, No. 2, 2004 amantadine twice daily, 5 mg selegiline three times daily, 4 mg trihexyphenidyl three times daily, and 5 mg bromocriptine three times daily. Examination in 1996 disclosed worsening of his dystonia. Although he still had no ophthalmic symptoms, he now had markedly abnormal eye movements. In straight-ahead gaze, his eyes were aligned and there were no saccadic intrusions. Pursuit was saccadic, as before. Horizontal gaze amplitude was normal, but both eyes displayed marked limitation of upward and downward gaze, estimated as 30% and 40% of normal range, respectively. Both upper lids were retracted. Saccades were slow in all directions; optokinetic nystagmus in the horizontal and vertical planes was symmetrical. Vestibulo- ocular reflexes by doll's eye maneuver were intact. The remainder of the ophthalmologic examination was normal. Ocular disturbances have not previously been reported in lubag, although their presence parallels those found in other movement disorders such as Parkinson disease and Huntington disease ( 5,6). In particular, the patient's eye movement abnormalities, with the primary exception of the absence of square wave jerks on primary gaze, bear a strong resemblance to those found in progressive supranuclear palsy ( 7). Although progressive supranuclear palsy neuropathology includes brainstem involvement ( 7), autopsy studies in lubag patients have shown atrophy of the caudate nucleus and putamen ( 2,8). In one case of lubag with myoclonus ( 9), it was suggested that extrastriatal structures may have been affected, but no other areas have specifically been implicated in the pathogenesis of this disease. In this case, the patient's marked supranuclear ocular disturbances appear consistent with brainstem deficits. Daniel D. Esmaili MD Department of Ophthalmology ( Doheny Eye Institute) University of Southern California Keck School of Medicine Los Angeles, California desmaili@ yahoo . com Richard A. Shubin MD Department of Neurology University of Southern California Keck School of Medicine Los Angeles, California Cheryl H. Waters, MD Department of Neurology Division of Movement Disorders Columbia University New York, New York Alfredo A. Sadun, MD, PhD Department of Ophthalmology ( Doheny Eye Institute) University of Southern California Keck School of Medicine Los Angeles, California REFERENCES 1. Lee LV, Pascasio FM, Fuentes FD, et al. Torsion dystonia in Panay, Philippines. Adv Neurol 1976; 14: 137- 51. 2. Lee LV, Munoz EL, Tan KT, et al. Sex linked recessive dystonia parkinsonism of Panay, Philippines ( XDP). Mol Pathol 2001; 54: 362- 8. 3. Lee LV, Kupke KG, Caballar- Gonzaga F, et al. The phenotype of the X- linked dystonia- parkinsonism syndrome: an assessment of 42 cases in the Philippines. Medicine 1991; 70: 179- 87. 4. Waters CH, Takahashi H, Wilhelmsen KC, et al. Phenotypic expression of X- linked dystonia- parkinsonism ( lubag) in two women. Neurology 1993; 43: 1555- 8. 5. Leigh RJ, Daroff RB, Troost BT. Supranuclear disorders of eye movements. In: Glaser JS ed. Neuro- ophthalmology, 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 1999: 351- 62. 6. Leigh RJ, Newman SA, Folstein SE, et al. Abnormal ocular motor control in Huntington's disease. Neurology 1983; 33: 1268- 75. 7. Steele JC, Richardson JC, Olszewski J. Progressive supranuclear palsy: A heterogeneous degeneration involving the brain stem, basal ganglia and cerebellum with vertical gaze and pseudobulbar palsy, nuchal dystonia and dementia. Arch Neurol 1964; 10: 333- 59. 8. Waters CH, Faust PL, Powers J, et al. Neuropathology of Lubag ( X- linked dystonia- parkinsonism). Mov Disord 1993; 8: 387- 90. 9. Evidente VG, Advincula J, Esteban R, et al. Phenomenology of " Lubag" or X- linked dystonia- parkinsonism. Mov Disord 2002; 17: 1271- 7. Laser Visual Field Testing To the Editor: In the recent issue of the journal, Michael Lee et al ( 1) reported the use of laser visual field testing for screening purposes. It is interesting to note that the authors used a red laser for their target. In the general male population, up to 8% may have red- green color blindness, and this might be a limitation of the testing in the subgroup. I wonder if any of the patients in their predominantly male Veterans Hospital cohort reported difficulty with the red laser target ( 2,3). I have also had anecdotal success using my laser pointer ( green) for testing visual fields in patients at the bedside, and I agree with the authors that the technique is useful, particularly when a formal visual field is not available or impractical. I commend the authors for their novel application of new technology in addressing an old problem. Andrew G. Lee, MD University of Iowa Hospitals and Clinics Iowa City, Iowa andrew- lee@ uiowa. edu 189 JNeuro- Ophthalmol, Vol. 24, No. 2, 2004 Letters to the Editor REFERENCES Lee MS, Baker LJ, Volpe NJ, et al. Laser pointer visual field screening. JNeuroophthalmol 2003; 23: 260- 3. Romano PE. Laser pointers and color blindness. Binoc Vision Strab Q 1999; 14: 7. Eagle RC. Laser pointers and color blindness. Ophthalmol 1998; 105: 760. Reply: We appreciate the comments of Dr. Andrew Lee. In our experience, patients who are red- color blind do not have trouble seeing a red laser pointer on a uniform testing background. We agree with Dr. Lee that a green laser pointer may also work for the purpose of screening. Michael S. Lee, MD Cole Eye Institute Cleveland Clinic Foundation Cleveland, Ohio leem4@ ccf. org Laura J. Baker, MD, MSc. E. Nicholas J. Volpe, MD Grant T. Liu, MD Gui S. Ying, MS Steven L. Galetta, MD Departments of Ophthalmology and Neurology University of Pennsylvania School of Medicine Philadelphia, Pennsylvania 190 © 2004 Lippincott Williams & Wilkins |