| Title |
Advanced Combustion Facilities and Diagnostics |
| Creator |
Dugué, Jacques; Drasek, W. Von; Samaniego, J. M.; Charon, O.; Oguro, T. |
| Publisher |
University of Utah |
| Date |
1998 |
| Spatial Coverage |
presented at Maui, Hawaii |
| Abstract |
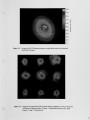
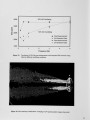
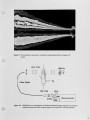
Air Liquide recently completed the commissioning of a new advanced furnace for combustion system development and testing. This flexible, large scale test platform located in France is designed for oxy-natural gas and oxy-heavy fuel oil flames with firing rates up to 2.0 MW. The new furnace is equipped with a continuous probe access slot and a fully automated 3-D traversing system allowing in-flame measurements in horizontal and vertical planes. The addition of this furnace expands Air Liquide's experimental facilities, which include 730 kW and 30 kW test furnaces at the Chicago Research Center. These furnaces can operate at wall temperatures of 1600°C and allow measurements of heat transfer profile, furnace thermal efficiency and flue gas compositions with a high level of accuracy and repeatability. This paper presents details of the new furnace and the associated flame diagnostics specifically designed for operating in high temperature oxy-flames. The conventional and laser measurement techniques routinely used at the Chicago and the Claude-Delorme Research Centers allow detailed flame characterization for gas composition, local or line-of-sight gas temperature, soot volume fraction. Other optical and imaging techniques are used for visualizing OH emission, characterizing burner jet mixing, and identifying spectral content of flame and furnace gases. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s6z03brb |
| Setname |
uu_afrc |
| ID |
10990 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s6z03brb |