| Title |
Lymphoplasmacyte-Rich Meningioma Mimicking Idiopathic Hypertrophic Pachymeningitis |
| Creator |
Hirunwiwatkul, P; Trobe, JD; Blaivas, M |
| Affiliation |
Department of Ophthalmology and Visual Sciences, University of Michigan, Ann Arbor, Michigan, USA. |
| Abstract |
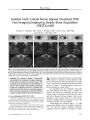
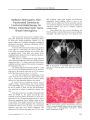
A 24-year-old man presented with long-term headache and progressive visual loss. Neuro-ophthalmic manifestations included finger counting acuity in both eyes, weakly reactive pupils, pale optic discs, and increased deep tendon reflexes. Brain MRI showed meningeal thickening that involved the optic nerves and chiasm and enveloped and displaced the brainstem as far caudally as the foramen magnum. The diffuse extensive nature of the lesion suggested an inflammatory process such as idiopathic hypertrophic pachymeningitis (IHP), but anterior temporal brain biopsy disclosed a relatively high proportion of meningothelial cells with islands of polyclonal inflammatory reaction consistent with a diagnosis of lymphoplasmacyte-rich meningioma (LRM), a rare variant. Among the 19 reported cases of LRM, none has shown as extensive a mass as seen in our patient. Distinguishing between LRM and IHP is important because these entities are treated differently. |
| Subject |
Adult; Diagnosis, Differential; Dura Mater, pathology; Humans; Hypertrophy; Magnetic Resonance Imaging; Male; Meningeal Neoplasms, diagnosis; Meningioma, diagnosis; Meningitis, diagnosis; Plasma Cells, pathology |
| Format |
application/pdf |
| Publication Type |
Journal Article |
| Collection |
Neuro-Ophthalmology Virtual Education Library: Journal of Neuro-Ophthalmology Archives: https://novel.utah.edu/jno/ |
| Publisher |
Lippincott, Williams & Wilkins |
| Holding Institution |
Spencer S. Eccles Health Sciences Library, University of Utah |
| Rights Management |
© North American Neuro-Ophthalmology Society |
| Setname |
ehsl_novel_jno |
| ID |
225642 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s60k5fn5/225642 |