| OCR Text |
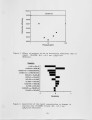
Show AFRC90 Paper #2 A CHEMICAL KINETIC MODELING STUDY OF CHLORINATED HYDROCARBON COMBUSTION William J . Pitz and Charles K. Westbrook Lawrence Livermore National Laboratory Livermore, CA 94550 ABSTRACT The combustion of chloroethane is modeled as a stirred reactor so that we can study critical emission characteristics of the reactor as a function of residence time. We examine important operating conditions such as pressure, temperature, and equivalence ratio and their influence on destructive efficiency of chloroethane. The model uses a detailed chemical kinetic mechanism that we have developed previously for C3 hydrocarbons. We have added to this mechanism the chemical kinetic mechanism for C2 chlorinated hydrocarbons developed by Senkan and coworkers . In the modeling calculations , sensitivity coefficients are determined to find which reaction-rate constants have the largest effect on destructive efficiency . INTRODUCTION Chlorinated hydrocarbons comprise a significant fraction of hazardous waste . Incineration is a commonly used method to dispose of these chlorinated hydrocarbons. Some of the drawbacks of incineration are that the hazardous component may not be completely destroyed, other hazardous components may be produced during the combustion process and both may be released as a pollutants . Release of these hazardous components can potentially cause an incinerator to exceed present or future emission requirements and can raise public concerns about the associated health risks . In this study, the chemical kinetics of the destruction of chlorinatea hydrocarbons are examined. The combustion process is modeled as a perfectly-stirred reactor with the inclusion of detailed chemical kinetics . This is a highly simplified physical model in which the hazardous component and oxidizer are ssumed to mix very rapidly with combustion products . However, this ~ eatment has an attractive feature . Practical combustors have highly turbulent regions where the chlorinated hydrocarbon is rapidly mixed with combustion products . These regions may be simulated as a stirred reactor with chemical kinetics controlling the extend of chlorinated hydrocarbon destruction and the production of any additional hazardous components . Lutz et al . [1] have used a turbulent model which includes two stirred reactors to model the production of pollutants in a turbulent jet. The numerical model considered allows the examination of a wide range of operating parameters such as temperature , pressure, residence time , and equivalence ratio. All these operating parameters are easily-specified , input parameters in a stirred reactor model . The model allows us to examine operating conditions not easily achieved in experimental studies , particularly high pressure . Our objective is to find conditions under which the maximum destruction of the chlorinated hydrocarbon is obtained and the minimum amount of other hazardous components are produced . -1- |