| Title |
NOx Reduction and Burnout Optimisation Using an Aerodynamically Air Staged Burner and an Air Staged Precombustor Burner |
| Creator |
Smart, J. P.; Weber, R. |
| Publisher |
University of Utah |
| Date |
1988 |
| Spatial Coverage |
presented at Pittsburgh, Pennsylvania |
| Abstract |
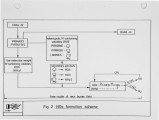
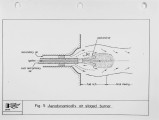
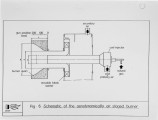
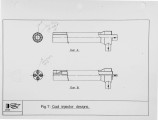
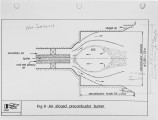
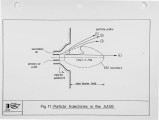
Recently, a significant body of work has been undertaken at the IFRF to develop low-NOx pulverised-coal burners. The majority of the work has focused on the design and evaluation of internal air staged burners at the 2.5 MW semi-i~dustrial scale. Specific emphasis has been given to retrofitting the burner designs to wall-fired combustion units. Two burner concepts have been evaluated, an aerodynamically air staged burner (AASB) and an air staged precombustor burner (ASPB). The dominant design feature of all air-staged burners is the creation of a fuel-rich zone in which volatile coal-nitrogen species react to molecular N2 in prefernce to NOx. The two burners create this fuel rich zone in fundamentally different ways. The AASB relies on optimisation of the near-burner field aerodynamics whereas the ASPB utilises a prechamber operating under sub-stoichiometric conditions. Both burner were fired with a high volatile and a medium volatile bituminous coal. Relatively low NOx emissions and acceptable burnout levels were achieved for both coals, however a coal type dependence was observed. Results of the two burners have been compared to an externally air staged burner firing the same coals. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s63f4s73 |
| Setname |
uu_afrc |
| ID |
5143 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s63f4s73 |