| OCR Text |
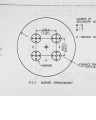
Show 2 (2) Promt NO : As the temperature-dependance is small and prompt NO is formed by participation of C and CH-radicals, it is effective to reduce the partial pressure of C and CH. For this purpose, fuel-lean or diluted combustion are effective. (3) Fuel NO : Since it is effective to reduce oxygen concentration, two-stage air combustion is generally employed. However, there is a limit to its effectiveness due to re-formation of NO from HCN, NH3, etc. by feeding of air to the second stage, and the emission problem of unburnt material. Other effective method is to decompose NO into N2 by decomposed gas such as NH3, CH, etc- in the downstream zone of the flame. Principle and Features : 3-1 Principle Fig. 1 shows an example of the structure of the two stage fuel burner. The principle is as follows. ."Divide fuel into the first and the second stage fuel and burn the first stage fuel rapidly with air necessary for combustion of the total fuel (primary combustion), and then burn the second stage fuel with the remaining oxygen in the gas after the primary combustion (secondary combustion)". The combustion process of the first stage fuel is important in this principle. NOx formation with regards to air ratio is generally as shown in Fig. 2. Namely, NOx formation decreases rapidly in the fuel richzone (air ratio is below 1.0), but in the fuel lean zone (air ratio is over 1.0) NOx formation differs largely depending on the type of combustion. In the premix combustion at high air ratio, formation of thermal NO and prompt NO is sufficiently controlled by the decrease of combustion temperature and the dilution of the fuel, so NOx is hardly formed. In our principle, the premix combustion is taken as a basis. However, in the case of liquid fuel, premixing is impossible because of the nature of the fuel. 1 0 -2 |