| OCR Text |
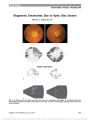
Show Literature Commentary Viola F, Villani E, Natacci F, Selicorni A, Melloni G, Vezzola D, Barteselli G, Mapelli C, Pirondini C, Ratiglia R. Choroidal abnormalities detected by near-infrared reflectance imaging as a new diagnostic criterion for neurofibromatosis 1. Ophthalmology. 2012;119:369-375. Objective: To investigate in a large sample of consecutive patients with neurofibromatosis type 1 (NF1) the possibility of including the presence of choroidal abnormalities detected by near-infrared reflectance (NIR) as a new di-agnostic criterion for NF1. Design: Cross-sectional evaluation of a diagnostic test. Participants and Controls: Ninety-five consecutive adult and pediatric patients (190 eyes) with NF1, diagnosed based on the National Institutes of Health (NIH) criteria. Controls in-cluded 100 healthy age- and gender-matched control subjects. Methods: Confocal scanning laser ophthalmoscopy was performed for each subject, investigating the presence and the number of choroidal abnormalities. Main Outcome Measures: Sensitivity, specificity, and di-agnostic accuracy for the different cutoff values of the criterion choroidal nodules detected by NIR compared with the NIH criteria. Results: Choroidal nodules detected by NIR imaging were present in 79 (82%) of 95 of the NF1 patients, including 15 (71%) of the 21 NF1 pediatric patients. Similar abnormalities were present in 7 (7%) of 100 healthy subjects, including 2 (8%) of the 25 healthy pediatric subjects. The highest accuracy was obtained at the cutoff value of 1.5 choroidal nodules detected by NIR imagery. Sensitivity and specificity of the examination at the optimal cutoff point were 83% and 96%, respectively. Diagnostic accuracy was 90% in the overall population and 83% in the pediatric population. Both of these values were in line with the most common NIH diagnostic criteria. Conclusions: Choroidal abnormalities appearing as bright patchy nodules detected by NIR imaging frequently occurred in NF1 patients. The present study shows that NIR examination to detect choroidal involvement should be considered as a new diagnostic criterion for NF1. The authors prospectively imaged fundi of patients with neurofibromatosis type 1 (NF1) and normal controls with near-infrared imaging using confocal scanning laser oph-thalmoscopy (cSLO). They identified choroidal nodules using cSLO in more than 80% of NF1 patients and 7% of controls. By comparison, Lisch nodules were detected in 72% of NF1 patients. The sensitivity (83%) and specificity (96%) of choroidal nodules in NF1 were high when at least 2 nodules were detected. These choroidal nodules are generally not visible using fundus examination, red-free photography, fluorescein angiography, or fundus autofluorescence. They can be seen using near-infrared imaging and indocyanine green angiography. I would encourage the reader to take a look at the images from the article because they are striking. The authors suggest that these lesions become a new diagnostic criterion for NF1, and I think it is reasonable given the high specificity. The noninvasive nature and rapid acquisition time of cSLO may improve the diagnostic accuracy when Lisch nodules are absent. -Michael S. Lee, MD The finding of choroidal nodules by noninvasive imaging will likely be helpful for the diagnosis of NF1. I agree it will be most useful when Lisch nodules are not seen, but we have to keep in mind that 7% of controls have 1 choroidal nodule and 4% have 2 nodules. Since the control group was normal subjects, we don't yet know whether other conditions might show similar findings. Nonetheless, I do think it will play a role in the future diagnosis of NF1. -Mark L. Moster, MD Menzel-Severing J, Siekmann U, Weinberger A, Roessler G, Walter P, Mazinani B. Early hyperbaric oxygen treatment for nonarteritic central retinal artery obstruction. Am J Ophthalmol. 2012;153:454-459. Purpose: To compare hyperbaric oxygen treatment com-bined with hemodilution with hemodilution only in central retinal artery obstruction. Design: Retrospective nonrandomized case series. Methods: We reviewed records of all our patients diagnosed with central retinal artery obstruction between 1997 and 2010. In these patients, hyperbaric oxygen and hemodilu-tion therapy had been administered routinely (oxygen group). Where hyperbaric oxygenation could not be per-formed, patients underwent hemodilution only (control group). Patients with presenting visual acuity (VA) of up to 20/200 within 12 hours of onset were included in our analysis. Exclusion criteria included cilioretinal vessels or arteritic occlusion. Results: The oxygen group comprised 51 patients, and the control group comprised 29 patients. Mean baseline VA was counting fingers (oxygen group) and 20/1000 (control group, P = 0.1). Most other potential confounders, including duration of symptoms, also did not differ significantly at baseline. In the oxygen group, mean VA improvement was Moster and Lee: J Neuro-Ophthalmol 2012; 32: 185-188 185 Literature Commentary Section Editors: Mark L. Moster, MD Michael S. Lee, MD Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. 3 lines (P , 0.0001). This was sustained over a follow-up of 3 months (P = 0.01). In the control group, mean improve-ment was 1 line (P = 0.23 at discharge; P = 0.17 at follow-up). Differences between both the groups were not signifi-cant (P = 0.07 at discharge; P = 0.26 at follow-up). The number of patients gaining 3 lines or more was 38.0% vs 17.9% at discharge (P = 0.06) and 35.7% vs 30.8% at follow-up (P = 0.76). Conclusions: We saw a significant VA improvement after the combined treatment but not when using hemodilution only. Confirming superiority of the combination treatment requires a randomized prospective trial. A high number of nonresponders highlight the need to improve our under-standing and treatment of hypoxia-related metabolic insults after central retinal artery obstruction. This retrospective chart review is from an institution whose routine treatment for central retinal artery obstruction (CRAO) has been a combination of hyperbaric oxygen (HBO) and hemodilution by hydration to a hematocrit ,40%. For various reasons, HBO was not given to 29 of 80 patients, who served as a control group. The study found a 3-line improvement from baseline with the combined treatment and a nonsignificant 1-line improvement in those with hemodilution alone. The study suffers from numerous limitations. First, there may be selection bias in that those not receiving HBO had the following reasons given: patient declined the procedure, the compression chamber was not avail-able, or there were systemic contraindications. Second, the control group had a near statistical (P = 0.06) increase in the history of coronary artery disease, stroke, or periph-eral vascular disease (16 of 29 vs 16 of 51) as well as in age (mean, 74 vs 69 years old). Third, there was no standard method of visual acuity measurement. Although HBO may truly benefit patients with CRAO, it is hard to determine from this study and, as the authors note, a prospective randomized clinical trial would be necessary. -Mark L. Moster, MD To me, the biggest issue represents the lack of masking and potential bias in checking visual acuity. The authors note that they proposed to treat everyone with HBO and the by-default control group consisted of those who could not have it. I think this suggests a lack of equipoise regarding the treatment. The patient and examiner knew who had HBO, and both may have worked harder to get the best visual acuity compared with controls. Finally, the percentage of patients who improved by 3 lines of acuity at discharge and at 3 months was no different between these groups. This does not support HBO as unequivocally beneficial especially in light of the potential bias. -Michael S. Lee, MD Ahmed RM, Wilkinson M, Parker GD, Thurtell MJ, Macdonald J, McCluskey PJ, Allan R, Dunne V, Hanlon M, Owler BK, Halmagyi GM. Transverse sinus stenting for idiopathic intracranial hypertension: a review of 52 patients and of model predictions. AJNR Am J Neuroradiol. 2011;32:1408-1414. Background and Purpose: Transverse sinus stenosis (TSS) is common in patients with idiopathic intracranial hyper-tension (IIH). While the role of TSS in IIH pathogenesis remains controversial, modeling studies suggest that stent placement within a TSS with a significant pressure gradient should decrease cerebral venous pressure, im-prove cerebrospinal fluid (CSF) resorption in the venous system, and thereby reduce intracranial (CSF) pressure, improving the symptoms of IIH and reducing papilledema. We aimed to determine if IIH could be reliably treated by stent placement in TSS. Materials And Methods: We reviewed the clinical, veno-graphic, and intracranial pressure data before and after stent placement in TSS in 52 of our own patients with IIH unres-ponsive to maximum acceptable medical treatment, treated since 2001 and followed between 2 months and 9 years. Results: Before stent placement, the mean superior sagit-tal sinus pressure was 34 mm Hg (462 mm H2O) with a mean TSS gradient of 20 mm Hg. The mean lumbar CSF pressure before stent placement was 322 mm H2O. In all 52 patients, stent placement immediately eliminated the TSS pressure gradient, rapidly improved IIH symptoms, and abolished papilledema. In 6 patients, symptom relapse (headache) was associated with increased venous pressure and recurrent stenosis adjacent to the previous stent. In these cases, placement of another stent again removed the TSS pressure gradient and improved symptoms. Of the 52 patients, 49 have been cured of all IIH symptoms. Conclusions: These findings indicate a role for transverse sinus stent placement in the management of selected patients with IIH. The authors stented an area of transverse sinus stenosis (TSS) in 52 patients with idiopathic intracranial hyper-tension (IIH) who had either failed medical therapy or suffered fulminant visual loss. The authors defined 2 different kinds of stenosis: extrinsic (smooth area of narrowing) or intrinsic (irregular area of narrowing due to an intraluminal band or granulation). Unfortunately, they did not differentiate whether the outcome was different between these 2 groups. Papilledema was present in 46 patients prestent and resolved in all. I wish the authors had noted what was the time interval between stenting and resolution of papille-dema. If it were within several weeks, this would suggest that the stent alone made the difference. If it were over several months and the patients remained on diuretics and lost weight, then it may not have made any difference. Interestingly, visual field loss was moderate or severe in only 11 patients. My practice is a bit more conservative and 186 Moster and Lee: J Neuro-Ophthalmol 2012; 32: 185-188 Literature Commentary Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. I would likely hold off on surgical intervention for the 41 patients with mild to no visual field loss. Finally, for those who could not attend, we heard, at the annual NANOS meeting in February 2012, that the presence of TSS did not affect the final outcome of patients with IIH. We also heard at a symposium that one group favoring stenting did so only if TSS did not resolve after lumbar puncture. Clearly, the role of venous sinus stenting in IIH remains unsettled. -Michael S. Lee, MD So the debate continues. Is stenosis of the transverse sinus the cause of IIH or a consequence of it? Is it the "chicken" or the "egg?" Those who argue against stenting because it may be the "egg" ignore the fact that none of our other treatments (diuretics, optic nerve sheath fenestration, lumbar or ventricular peritoneal shunt) address the under-lying cause of IIH. The results of this study are impressive and demonstrate that TSS for IIH is effective. The study is retrospective and without a standardized protocol for vision testing. Nonethe-less, every feature of symptomatic history and physical exam-ination was improved in this patient population after TSS. An important issue is the risk of complications of the treatment. The authors point out that shunts and optic nerve sheath fenestration have high complication rates as well. With TSS, 2 of 52 patients had serious complications. One had a subdural hematoma and 1 subdural, subarachnoid, and intracerebral bleeding. Both made a full recovery after craniotomy. Although a low overall rate of complications, the type of complication is quite severe and concerning. I do agree with Michael that I would reserve any surgical treatment for patients with progressive visual acuity and field loss and many in the current study would not meet those criteria. Many patients were treated who had no visual loss. Where will TSS fit in the treatment of IIH? It will likely vary by institution. Some centers with highly developed vascular interventional expertise may use it earlier in the course of IIH. In other centers, it may play a role in patients with progressive symptoms who fail other interventions.Wouldn't it be nice to have a prospec-tive, randomized .. -Mark L. Moster, MD Postoperative Visual Loss Study Group. Risk factors associated with ischemic optic neuropathy after spinal fusion surgery. Anesthesiology. 2012;116:15-24. Background: Perioperative visual loss, a rare but dreaded complication of spinal fusion surgery, is most commonly caused by ischemic optic neuropathy (ION). The authors sought to determine the risk factors for ION in this setting. Methods: Using a multicenter case-control design, the authors compared 80 adult patients with ION from the American Society of Anesthesiologists Postoperative Visual Loss Registry with 315 adult control subjects without ION after spinal fusion surgery, randomly selected from 17 institutions, and matched by the year of surgery. Preexisting medical conditions and perioperative factors were compared between patients and control subjects using stepwise multivariate analysis to assess factors that might predict ION. Results: After multivariate analysis, risk factors for ION after spinal fusion surgery included male sex (odds ratio [OR], 2.53; 95% confidence interval [CI], 1.35-4.91; P = 0.005), obesity (OR, 2.83; 95% CI, 1.52-5.39; P = 0.001), Wilson frame use (OR, 4.30; 95% CI, 2.13-8.75; P , 0.001), anes-thesia duration (OR per 1 hour = 1.39; 95% CI, 1.22-1.58; P , 0.001), estimated blood loss (OR per 1 L = 1.34; 95% CI, 1.13-1.61; P = 0.001), and colloid as percent of non-blood replacement (OR per 5% = 0.67; 95% CI, 0.52-0.82; P , 0.001). After cross-validation, area under the curve was 0.85, sensitivity was 0.79, and specificity was 0.82. Conclusions: This is the first study to assess ION risk factors in a large, multicenter, case-control fashion with detailed perioperative data. Obesity, male sex, Wilson frame use, longer anesthetic duration, greater estimated blood loss, and decreased percent colloid administration were significantly and independently associated with ION after spinal fusion surgery. Visual loss from ischemic optic neuropathy (ION) is a devastating complication of spinal surgery. Prior studies have estimated an occurrence between 0.017% to 0.1% and have suggested that risk factors include anemia, hypoten-sion, blood loss, large fluid shifts, venous congestion of the orbits, and coexisting diseases, such as atherosclerotic vascular disease, diabetes, obesity, and hypertension. How-ever, these past studies have limited value. They report either small numbers of patients from single institutions or data from national inpatient databases, which lack detailed perioperative data. Additionally, these same risk factors exist in many patients who did not develop ION. The current case-control study of 80 patients from the American Society of Anesthesiologists Postoperative Visual Loss Registry provides the best information to date. It found the following independent risk factors for ION: male sex, obesity, Wilson frame use, longer anesthetic duration, greater estimated blood loss, and lower percent colloid administration. Interest-ingly, other vasculopathic risk factors (older age, hypertension, smoking) were not independent risk factors and suggest that the cause of ION may be influenced more by the intraoperative physiologic changes than by preexisting diseases. The study moves us forward to a possibility of prevent-ing ION by modifying some of the risk factors, avoiding the Wilson frame, minimizing duration of anesthesia, and limiting blood loss. -Mark L. Moster, MD I think it is important to note that this refers to spine surgery only and may not apply to other surgical cases of Moster and Lee: J Neuro-Ophthalmol 2012; 32: 185-188 187 Literature Commentary Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. postoperative vision loss. It is also important to note that the data from the controls was more rigorously collected than the patients with ischemic optic neuropathy, and there may be some other risk factor as yet unidentified. Finally, the authors acknowledge that staging the procedure and using more colloid may have other deleterious effects. A risk-benefit analysis of modifying these factors is warranted. -Michael S. Lee, MD Raser JM, Mullen MT, Kasner SE, Cucchiara BL, Messé SR. Cervical carotid artery dissection is associated with styloid process length. Neurology. 2011;77:2061-2066. Objective: To investigate whether cervical carotid artery dissection (CCAD) is associated with anatomic character-istics of the styloid process of the temporal bone. Methods: CT angiograms from 38 case patients with CCAD and 38 age- and sex-matched control subjects without dissection were studied. Styloid process length, angulation in the axial and rostral-caudal planes, and proximity to the carotid artery were measured bilaterally by raters blinded to radiology reports and clinical information. Results: In control subjects, there was substantial variation in styloid process length (range, 4.6-42.9 mm), medial an-gulation (range, 16-89°), caudal angulation (range, 31-80°), and proximity to the carotid (range, 0.7-15.4 mm). Control subjects also demonstrated marked symmetry between sides (correlation coefficients: 0.80 for length, 0.34 for proximity, 0.81 for medial angulation, and 0.87 for caudal angulation). In case patients, the mean styloid length on the side of the dissection was not significantly longer than that on the con-tralateral side (mean, 30.3 vs 29.7 mm; P = 0.30). The styloid process was significantly longer ipsilateral to the dis-section in case patients compared with the side-matched pro-cess in control subjects (mean, 30.3 vs 26.6 mm; P = 0.03). Carotid dissection was associated with increasing styloid pro-cess length (odds ratio [OR], 1.08/mm; 95% confidence in-terval [CI], 1.002-1.17; P = 0.04). The OR of dissection in the highest quartile of length compared with that in the lower 3 quartiles was 4.0 (95% CI, 1.3-14.2; P = 0.03). Conclusions: CCAD is associated with a longer styloid process, suggesting that mechanical injury from the styloid may contribute to the pathogenesis of CCAD. The cause of cervical carotid artery dissection (CCAD) is often uncertain. This study was performed in part because of prior rare case reports of elongated styloid process with CCAD and carotid compression with ischemia. The main finding is a longer styloid process in patients with CCAD compared with the ipsilateral side of controls without dissection. Since the ipsilateral carotid lumen was distorted in the CCAD patients, the authors studied the proximity of the styloid process to the carotid artery contralaterally and found that the styloid process was closer to the carotid artery than in controls. This is a preliminary retrospective case-control study, but its findings suggests that mechanical injury from an elongated styloid process may contribute to CCAD and make damage from even seemingly minor injuries more plausible. -Mark L. Moster, MD Eighty-four percent of the carotid dissections occurred within the carotid region adjacent to the styloid process, further bolstering the plausibility of the association. There are a couple of major limitations that may eventually undo this hypothesis. The authors could not have been masked to cases vs controls since they could see the dissection on the scans. This could lead to bias in the measurements regarding proximity and styloid process length. Second, although the mean styloid process length was longer among cases than controls, the ranges were very broad (5-43 mm!). The mean values may have been statistically different, but is it really clinically significant since the overlap was so great? -Michael S. Lee, MD Bastawrous A. Smartphone fundoscopy. Ophthalmology. 2012;119:432-433. This was a letter to the editor in Ophthalmology, but the content was intriguing. We have all been contacted by telephone about inpatient or emergency room consults where it would have been nice to see the fundus. As more and more residents and fellows carry smartphones, this letter describes using a smartphone to image the fundus. The author turned on the flash while using the video mode to provide a constant source of coaxial illumination. The examiner then held the 20-diopter lens in the normal fashion for a dilated fundus examination in one hand and the smartphone in the other hand. The smartphone has the ability to focus the image and also zoom in as necessary. After the recording has finished, the user can save a still image from the smartphone. The figures make it look relatively easy, and the fundus image quality appears reasonable. -Michael S. Lee, MD Michael, I looked at the image posted online with this letter to the editor and it seems the image is barely "reason-able." I tried it on 2 patients today and was only able to get a narrow retinal image with good quality. Zoom is not avail-able in video mode, so the magnification is based on moving the lens and smartphone as well as in computer processing after acquiring the image, which decreases the resolution. Although with me, it is a "not so smart" phone fundoscopy, I'd say this is a good start and there will soon be better ways to acquire fundus photographs with our phones. Stay tuned! -Mark L. Moster, MD 188 Moster and Lee: J Neuro-Ophthalmol 2012; 32: 185-188 Literature Commentary Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. [VBneurofibromatosisone] |