| OCR Text |
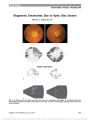
Show [CFchiasmischemia] Chiasmal Stroke in Patient With Atrial Fibrillation and Complete Occlusion of Right Internal Carotid Artery Fabian et al (1) recently published a case of "Chiasmal stroke following open-heart surgery" in the Journal, which described a 62-year-old man with total loss of vision in his right eye and loss of the temporal half of the vision in the left eye, and an otherwise normal neurologic examination. We report another case of chiasmal stroke. A 59-year-old man with probable myocarditis and a several month history of intermittent shortness of breath was diagnosed with atrial fibrillation with rapid ventricular rate and started on anticoagulation. Eight days later he experienced sudden complete vision loss in the right eye and loss of the temporal half of the visual field in the left eye. He was found to have an atrial thrombus and complete occlusion of his right internal carotid artery. Three days after acute vision loss, visual acuity was of no light perception, right eye, and 20/30, left eye. The right pupil was amaurotic. All other aspects of the examination, including appearance of the optic nerves, were normal. Automated perimetry demonstrated complete temporal field loss in the left eye respecting the vertical median (Fig. 1). His neurologic examination was normal. The day after vision loss, MRI of the brain showed an abnormality in the anterior chiasm on the right (Fig. 2). In addition, there was occlusion of the right internal carotid artery, an acute small left cerebellar infarct, and white matter changes consistent with small vessel ischemia. This patient had normal laboratory values for rapid plasma reagin and anti-neutrophil cytoplasmic antibody. Acute chiasmal infarction is a rare event, presumably because of its redundant and complex blood supply. To date, the only other report of chiasmal infarct was in a patient with acquired immunodeficiency syndrome, mucormyosis, and diabetes mellitus (2). We believe that the cause of our patient's infarction was stroke, with embo-lus from a left atrial thrombus. Julie B. Shelton, MD John A Moran Eye Center, Department of Ophthalmology and Visual Sciences, University of Utah, Salt Lake City Department of Neurology, University of Utah Clinical Neurosciences Center, Salt Lake City Kellogg Eye Center, Department of Ophthalmology University of Michigan, Ann Arbor Kathleen B. Digre, MD Bradley J. Katz, MD, PhD Judith E. A. Warner, MD John A Moran Eye Center, Department of Ophthalmology and Visual Sciences, University of Utah, Salt Lake City Department of Neurology, University of Utah Clinical Neurosciences Center, Salt Lake City Edward P. Quigley, MD, PhD Neuroradiology Section, Department of Radiology University of Utah School of Medicine, Salt Lake City julieshe@med.umich.edu Supported in part by an unrestricted grant to the Department of Ophthalmology from the Research to Prevent Blindness, Inc, New York, NY. The authors report no conflicts of interest. REFERENCES 1. Fabian ID, Greenberg G, Huna-Baron R. Chiasmal stroke following open-heart surgery. J Neuroophthalmol. 2010;30:219-221. 2. Lee BL, Holland GN, Glasgow BJ. Chiasmal infarction and sudden blindness caused by mucormycosis in AIDS and diabetes mellitus. Am J Ophthalmol. 1996;122:895-896. FIG. 1. Complete temporal visual field loss is present in the left eye. FIG. 2. Chiasmal infarction. A. Diffusion-weighted imaging shows bright signal (arrowhead ) in the anterior chiasm on the right and apparent diffusion coefficient map (not shown ) confirmed restricted diffusion. B. Coronal fluid-attenuated inversion recovery (FLAIR) image reveals a signal abnor-mality (arrow ) in the same location. Letters to the Editor: J Neuro-Ophthalmol 2012; 32: 189-193 189 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. Aminopyridine Treatment in a Patient With Bilateral Vestibular Failure and Cryptogenic Downbeat Nystagmus In a recent issue of this journal, Kalla et al (1) reported that 4-aminopyridine (4-AP) alleviates the symptoms of downbeat nystagmus (DBN) significantly more than 3,4- diaminopyridine (3,4-DAP). Both drugs are potassium-channel blockers, but 4-aminopyridine is lipid-soluble and more easily crosses the blood-brain-barrier. 3,4-diaminopyr-idine is not lipid soluble, with direct effects in the peripheral nervous system and indirect effects in the central nervous system via its metabolites (1,2). In the report by Kalla et al (1), 1 male patient suffered from bilateral vestibular failure (disorder of the peripheral nervous system) as well as DBN (disorder of the central nervous system). We now document this case in detail. At the time of the study, the patient was 58 years old. Ten years earlier, he had traveled to Indonesia, where he suffered from extreme diarrhea with severe weight loss, followed by the development of tinnitus, oscillopsia, postural instability, and bilateral vestibular failure. MRI of the brain was normal. On examination, he was found to have DBN, which was documented with electronystagmog-raphy. The patient showed improvement of DBN following the administration of 3,4-DAP but not with 4-AP. The reason for this finding might be that the effects of 3,4-DAP are more balanced between the peripheral and the central nervous systems, while the effects of 4-AP are primarily on the central nervous system. It had been suggested that the combination of bilateral vestibular failure and idiopathic DBN is due to a multisystem channelopathy, decreasing the calcium currents through P/Q channels and thus impacting potassium channels both in the peripheral and the central nervous systems (3-6). This multisystem channelopathy could explain disorders not only in the peripheral nervous system (such as bilateral vestibular failure) but also in the central nervous system (such as DBN). 3,4-DAP may restore the excitability of the cerebellar Purkinje cells at a similar pace as it restores peripheral functioning, hence stabilizing the feedback loop between their central effect on eye movements and their interaction with peripheral structures responsible for balance. Rainer Spiegel, PhD Department of Neurology and IFBLMU Munich University Hospital, Campus Großhadern Munich, Germany Roger Kalla, MD Jens Classen, MD Stanislavs Bardins, MSc Fábio Anciães da Silva, MD Parvis Farahmand, MD Ales Hahn, MD Erich Schneider, PhD Nicole Rettinger, CO Klaus Jahn, MD Thomas Brandt, MD, FRCP Michael Strupp, MD Integrated Center for Research and Treatment of Vertigo Balance and Ocular Motor Disorders University of Munich Hospital Munich, Germany rainer.spiegel@med.uni-muenchen.de The authors report no conflict of interest. REFERENCES 1. Kalla R, Spiegel R, Claassen J, Bardins S, Hahn A, Schneider E, Rettinger N, Glasauer S, Brandt T, Strupp M. Comparison of 10 mg doses of 4-aminopyridine and 3,4-diaminopyridine for the treatment of downbeat nystagmus. J Neuroophthalmol. 2011;31:320-325. 2. Judge SIV, Bever CT Jr. Potassium channel blockers in multiple sclerosis: Neuronal Kv channels and effects of symptomatic treatment. Pharmacol Ther. 2006;111:224-259. 3. Wagner JN, Glaser M, Brandt T, Strupp M. Downbeat nystagmus: aetiology and comorbidity in 117 patients. J Neurol Neurosurg Psychiatry. 2008;79:672-677. 4. Stahl JS. Eye movements of the murine P/Q calcium channel mutant rocker, and the impact of aging. J Neurophysiol. 2004;91:2066-2078. 5. Stahl JS, James RA. Neural integrator function in murine CACNA1A mutants. Ann N Y Acad Sci. 2005;1039:580-582. 6. Womack MD, Chevez C, Khodakhah K. Calcium-activated potassium channels are selectively coupled to P/Q-type calcium channels in cerebellar Purkinje neurons. J Neurosci. 2004;24:8818-8822. In Memoriam, Noble J. David, MD: A Comment I was moved by the tribute to Noble J. David by Dr Lopez. I would like to add to the lore of Dr David-he was in command of all of the lyrics to the Gilbert and 190 Letters to the Editor: J Neuro-Ophthalmol 2012; 32: 189-193 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. Sullivan Operas (with which he entertained colleagues and me on numerous occasions). And he was a skilled speaker and raconteur, but I had a special love of our cello-piano sessions. He called himself, in contrast with the usual category of child prodigy, an "adult non-prodigy." However, our experiences in the cello-piano genre were very enjoyable-he introduced me to Paul Hindemith's "Drei Leichte Stücke" (3 easy pieces) for cello and piano, as well as more of the standards. He was definitely one of a kind, and we all shall miss him. Richard Sogg, MD Emeritus at Stanford University School of Medicine Los Gatos, California rlsogg@yahoo.com The author reports no conflicts of interest. Periodic Unilateral Eyelid Retraction in a Pediatric Patient: Some Considerations on Botulinum Toxin as a Temporary Treatment Option We read with great interest the recent case report by Gandhi et al (1), and specifically on the management of congenital eyelid retraction, we have the following com-ments to offer. Although surgical repair adopted by Gandhi et al (1) has been described as the main therapeutic approach for congenital eyelid retraction (2), this procedure involves risks and the outcomes are definitive and unpredictable (3). In this context, we (4) recently proposed a new non-surgical procedure for the treatment of congenital upper eyelid retraction based on the treatment of upper lid re-traction of Graves ophthalmopathy. Botulinum toxin type A was injected transcutaneously aiming at the complex of levator aponeurosis and Müller muscle. Optimal aesthetic and functional outcomes were obtained 10 days after the application with no side effects. Even as a temporary treatment option, especially those patients who refuse to undergo surgical repair may benefit from this approach. Furthermore, we believe that con-genital upper eyelid retraction patients (or their physi-cians) should be informed on the existence of alternative therapeutic methods (4,5) and on risks and benefits of existing therapeutic approaches (2-4) before any surgical intervention. Rafael Denadai, MD Érika Malheiros Bastos, MD, PhD Division of Plastic and Reconstructive Surgery Department of Surgery, School of Medical Sciences University of Marília, Marília São Paulo, Brazil The authors report no conflicts of interest. REFERENCES 1. Gandhi NG, Rogers GM, Kardon RH, Allen RC. Periodic unilateral eyelid retraction in a pediatric patient. J Neuroophthalmol. 2011;31:350-352. 2. Katowitz WR, Katowitz JA. Congenital and developmental eyelid abnormalities. Plast Reconstr Surg. 2009;124:93e-105e. 3. Tucker SM, Collin R. Repair of upper eyelid retraction: a comparison between adjustable and non-adjustable sutures. Br J Ophthalmol. 1995;79:658-660. 4. Bastos EM, Silva RD. Injection of transcutaneous botulinum toxin as a temporary treatment option for congenital eyelid retraction. Eur J Plast Surg. 2012;35:317-319. 5. Senyuva C, Yücel A, Yildirim I. Congenital upper eyelid retraction treated with gold weight implantation. Plast Reconstr Surg. 1997;99:2099-2100. Periodic Unilateral Eyelid Retraction in a Pediatric Patient: Comment and Response We appreciate the interest of Drs Denadai and Bastos in our case report as they propose the use of botulinum toxin in the treatment of congenital eyelid retraction (1). Others (2-8) have also provided evidence of nonincisional techniques for the treatment of upper eyelid retraction, most commonly secondary to thyroid eye disease, using botulinum toxin or dermal fillers. Currently, the preferred surgical treatment of upper eyelid retraction is the full-thickness blepharotomy, which has proven to be safe, effective, and predictable in experienced hands (9-12). This is the technique that our Letters to the Editor: J Neuro-Ophthalmol 2012; 32: 189-193 191 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. patient underwent. Advantages include a finite number of patient visits and a single procedure. Disadvantages include all potential complications of eyelid surgery. In addition, the etiology of the eyelid retraction in our patient would never have been determined if we had not placed her under anesthesia! The advantages of botulinum toxin injection include topical or no anesthesia and a lack of the potential complications associated with surgery. One disadvantage of using botulinum toxin is potential under- and over-correction due to variable diffusion patterns of the toxin based on the placement of the needle. Another disadvan-tage is indefinite number of office visits due to the temporary effect of the toxin (2.5 months in the case described by Drs Denadai and Bastos) and the inconve-nience of repeated injections. Comparative cost of the 2 procedures is an interesting issue. This would be a finite amount for a one time surgical procedure but would certainly be higher with repeated injections of botulinum toxin over months to years. We submit that in a patient who is a good surgical candidate, a full-thickness blepharotomy performed by an experienced surgeon is preferred over repeated botulinum toxin injections for upper eyelid retraction. We agree that all advantages, disadvantages, risks, and benefits of the treatment options should be fully disclosed to the patient. Nandini G. Gandhi, MD Richard C. Allen, MD, PhD UC Davis Eye Center Sacramento, California The authors report no conflicts of interest. REFERENCES 1. Bastos EM, Silva RDP. Injection of transcutaneous botulinum toxin as a temporary treatment option for congenital eyelid retraction. Eur J Plast Surg. 2011; doi:10.1007/s00238-011- 0570-5. 2. Biglan AW. Control of eyelid retraction associated with Graves' disease with botulinum A toxin. Ophthalmic Surg. 1994;25:186-188. 3. Uddin JM, Davis PD. Treatment of upper eyelid retraction associated with thyroid eye disease with subconjunctival botulinum toxin injection. Ophthalmology. 2002;109: 1183-1187. 4. Shih MJ, Liao SL, Lu HY. A single transcutaneous injection with Botox for dysthyroid lid retraction. Eye. 2004;18:466-469. 5. Costa PG, Saraiva FP, Pereira IC, Monteiro ML, Matayoshi S. Comparative study of Botox injection for upper eyelid retraction with 6-month follow-up in patients with thyroid eye disease in the congestive or fibrotic stage. Eye. 2009;23:767-773. 6. Mancini R, Khadavi NM, Goldberg RA. Nonsurgical management of upper eyelid margin asymmetry using hyaluronic acid gel filler. Ophthal Plast Reconstr Surg. 2011;27:1-3. 7. Salour H, Bagheri B, Aletaha M, Babsharif B, Kleshadi M, Abrishami M, Bagheri A. Transcutaneous Dysport injection for treatment of upper eyelid retraction associated with thyroid eye disease. Orbit. 2010;29:114-118. 8. Vasquez LM, Gonzalez-Candial M. Hyaluronic acid treatment for upper eyelid retraction after glaucoma filtering surgery. Orbit. 2011;30:16-17. 9. Elner VM, Hassan AS, Frueh BR. Graded full-thickness anterior blepharotomy for upper eyelid retraction. Arch Ophthalmol. 2004;122:55-60. 10. Hintschich C, Haritoglou C. Full thickness eyelid transsection (blepharotomy) for upper eyelid lengthening in lid retraction associated with Graves' disease. Br J Ophthalmol. 2005;89:413-416. 11. Demirci H, Hassan AS, Reck SD, Frueh BR, Elner VM. Graded full-thickness anterior blepharotomy for correction of upper eyelid retraction not associated with thyroid eye disease. Ophthal Plast Reconstr Surg. 2007;23:39-45. 12. Kazim M, Gold KG. A review of surgical techniques to correct upper eyelid retraction associated with thyroid eye disease. Curr Opin Ophthalmol. 2011;22:391-393. How Do We Define a Neuro-Ophthalmologist? I am writing to challenge a sentence in Byron Lam's tribute to J. Lawton Smith and Joel S. Glaser (1) in which he states that in 1962 there were only 7 Neuro-ophthalmologists in the world. The first question is how do we define a neuro-ophthalmologist? Of 2 greats at that time, David G. Cogan spent less than 10% of his time seeing patients. Frank B. Walsh spent no more than 30% of his time seeing neuro-ophthalmic patients. The rest was a general, consultative ophthalmic practice. Should we define a neuro-ophthalmologist as a practitioner who has had a fellowship before beginning practice? Should we differentiate between neuro-ophthalmologists and oph-thalmic neurologists? Should we include anyone who contributed understanding of neuro-ophthalmic entities? The following is a list of 34 physicians who were practicing some level of neuro-ophthalmology in 1962. It • Henderson Almeida • Jean Real Brunette • William McDonald Boles • Frank Carroll • David G. Cogan • James "Barry" Cullen • Andy Gay • Jack Goldstein • Angela Guido • David Harrington • Thomas R. Hedges, Jr • John W. Henderson • Robert Hollenhorst • William F. Hoyt 192 Letters to the Editor: J Neuro-Ophthalmol 2012; 32: 189-193 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. reflects my best "memory," and I apologize for those of whom I may have overlooked. David L. Knox, MD Wilmer Eye Institute The Johns Hopkins School of Medicine Baltimore, Maryland daknox@jhmi.edu REFERENCE 1. Lam BL. Tribute to J. Lawton Smith and Joel S. Glaser. J Neuroophthalmol. 2011;31:294-295. Insight Into 8 Patients With Nonarteritic Anterior Ischemic Optic Neuropathy Following Anti-VEGF Injections We read with great interest the recent article by Prescott et al (1) concluding that anti-vascular endothelial growth factor (anti-VEGF) therapy has no apparent bene-ficial effect in the treatment of nonarteritic anterior ischemic optic neuropathy (NAION). We would add a fur-ther note of caution in the use of anti-VEGF agents. Re-cently, we reported that these agents may actually compromise ocular blood flow and may precipitate an epi-sode of NAION (2). We described 8 patients who received intravitreal injection of one of these medications (bevacizu-mab, ranibizumab, pegaptnib) and developed NAION within 4-60 days. Because our patient cohort was small and the study design was retrospective, we cannot determine whether a causal relationship exists. Potential mechanisms include the vasoconstrictor effect of the anti-VEGF drugs, an increase in intraocular pressure from the intravitreal injection, and the exacerbation of systemic hypertension from the stress of the procedure. Further research is necessary to explore the potential relationship between intravitreal anti- VEGF therapy and NAION. Ahmad M. Mansour, MD Department of Ophthalmology, American University of Beirut Beirut, Lebanon Rafic Hariri University Hospital Beirut, Lebanon ammansourmd@gmail.com Stephen G. Schwartz, MD, MBA Ninel Z. Gregori, MD Zohar Yehoshua, MD, MHA Bascom Palmer Eye Institute, University of Miami Miller School of Medicine, Miami, Florida K. Bailey Freund, MD Kara Dellatorre, MD Vitreous-Retina-Macula Consultants of New York New York, New York Sabine Aisenbrey, MD University of Tübingen, Tübingen, Germany Hamid Hosseini, MD Khalili Hospital, Shiraz, Iran Jane Y. Huang, MD Fukuoka University, Fukuoka, Japan Ali A. Bodla, MD Birmingham and Midland Eye Centre Birmingham, United Kingdom M. Tariq Bhatti, MD Duke University Eye Center, Duke University Medical Center Durham, North Carolina The author reports no conflicts of interest. REFERENCES 1. Prescott CR, Sklar CA, Lesser RL, Adelman RA. Is intravitreal bevacizumab an effective treatment option for nonarteritic anterior ischemic optic neuropathy? J Neuroophthalmol. 2012;13:51-53. 2. Mansour AM, Shahin M. Kofoed PK, Parodi MB, Shami M, Schwartz SG. Insight into 144 patients with ocular vascular events during VEGF antagonist injections. Clin Ophthalmol. 2012;343-363. • Freddie Huber • Jamshed Juania • Timoteo Jimenez • Thomas Kearns • Alfred Kestenbaum • David L. Knox • Lois Lloyd • Irene Lowenfeld • Otto Lowenstein • J. Donald Lyle • Frank McAuley • Alfred McKinna • Swithin Meadows • Steven Miller • Adolph Neetens • Edward D. Norton • Charles Regan • C. Wilbur Rucker • Nathan Schlessinger • J. Lawton Smith • Frank B. Walsh Letters to the Editor: J Neuro-Ophthalmol 2012; 32: 189-193 193 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |