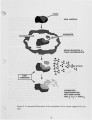
| OCR Text |
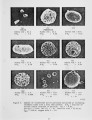
Show experiment. The error analysis also revealed that the largest uncertainty in the data comes from the sample to sample variation in iron content rather than experimental or analysis errors. Similar results were obtained for the smaller coal particles. These are illustrated in Figure 3 The time scale for the heat-up, devolatilization, and oxygen attack of these smaller particles is shorter due to their smaller mass. This is reflected in the data. For example, at 47 ms residence time the larger particles had not lost any iron whereas these smaller particles show an appreciable loss in iron. These smaller particles burned at slightly lower temperatures (~ 1600K) than their larger counterparts. This lower temperature, coupled with the smaller diameter, led to larger penetration of oxygen into the particle interior. The iron release mechanism shows a sensitivity to this larger penetration. That is, the ultimate extent of iron loss is larger when the penetration of oxygen into the particle interior increases. This trend underscores the sensitivity of the iron release mechanism to oxygen. The opposite trend would be anticipated had the mechanism of iron release been vaporization. The solid samples were also submitted to an automated image analysis technique which utilizes the energy dispersive spectroscopy probe of a scanning electron microscope. This technique performs a particle-by-particle analysis of the minerals in the sample and calculates their composition and size distributions. Approximately 600 particles are included in each analysis. The results of this analysis showed that the iron evolution occurred after pyrite had decomposed. For example, there was little pyrite remaining in the char at a residence time of 47 ms, even though there was no detectable amount of iron loss from the large particles. The analysis also showed that the pyrites did fracture during heatup, forming particles with roughly half the diameter of the original particles. The smaller particles were also observed to participate preferentially in the iron release. The original pyrite grains in the coal had a mass mean diameter of about 10 J1-m . The iron lost from these samples dominated the total release of inorganic elements. Figure 4 illustrates the release of all elements from the char on a mass basis. This figure is based on the largest initial coal particles in the 6 % oxygen environment. In the case of the smaller particles, where the fractional release of iron increased, the amount of iron released was more dominant still. In all cases, more than 80 % of the mineral matter released from these char particles (excluding sulfur) was iron. Coal particles containing a high percentage of pyritic iron were also entrained in a laminar flow reactor with electrically heated, inert gases. This reactor included similar in situ optical measurements of particle size , velocity, and temperature. No iron loss was observed from these sam pIes. This result helps explain the lack of pyrite 11 |