| OCR Text |
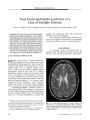
Show VIEWPOINT Rethinking Neuromyelitis Optica ( Devic Disease) Shelley Ann Cross, MD Abstract: Neuromyelitis optica ( NMO), or Devic disease, has been distinguished from multiple sclerosis ( MS) by the presence of optic neuritis that is usually bilateral, simultaneous, and often severe, myelopathic findings accompanied by longitudinally extensive spinal cord imaging abnormalities, no brain imaging abnormalities typical of MS, and often rapid progression to debility and even death. Researchers at the Mayo Clinic have identified an immunoglobulin marker of NMO ( the " NMO antibody") that binds selectively to the aquaphorin- 4 water channel and may play a causative role. This marker has been found in Japanese patients with opticospinal MS, prompting the suggestion that NMO and Japanese opticospinal MS are the same disorder. The NMO antibody, which predicts frequent relapse of myelopathy and optic neuritis, is also found in patients with lupus erythematosus and Sjogren syndrome who also have severe optic neuritis and longitudinally extensive myelitis. Because this antibody is also found in patients with optic neuritis and myelitis who have brain signal abnormalities atypical of MS, the diagnosis of NMO has been revised to allow inclusion of these brain imaging abnormalities. Proper distinction of NMO from MS is important because the two disorders may respond differently to immune modulatory therapy. (/ Neuro- Ophthalmol 2007; 27: 57- 60) The distinction between neuromyelitis optica ( NMO), or Devic disease, and multiple sclerosis ( MS) has long been unclear. Traditionally, NMO is believed to differ from MS by causing very severe, often bilateral, optic neuritis and longitudinally extensive MRI spinal cord lesions but no MRI brain lesions and aggressive progression to debility and death. In the past 7 years, investigators at the Mayo Clinic have made an effort to delineate objective criteria to Department of Neurology, Mayo Clinic, Rochester, Minnesota. Address correspondence to Shelley Ann Cross, MD, Department of Neurology, Mayo Clinic, 200 SW First Street, Rochester, MN 55905; E- mail: shelley. cross@ mayo. edu distinguish NMO from MS. Their efforts have resulted in a proposed redefinition of the diagnostic criteria. THE THREE PHENOTYPIC FORMS OF NEUROMYELITIS OPTICA The NMO phenotype occurs in three contexts: 1. MS. The optic nerves and spinal cord are the principal or only targets. MRI studies show scattered and also periventricular brain lesions and patchy and peripheral rather than central and longitudinally extensive spinal cord lesions. Oligoclonal bands are present in the cerebrospinal fluid ( CSF). The pathology consists of demyelination but generally not necrosis. 2. True NMO. Patients have recurrent severe optic neuritis and myelitis with longitudinally extensive MRI spinal cord lesions. There are no CSF oligoclonal bands. The pathology shows necrosis. Japanese and African- American patients with " opticospinal MS" may belong in this group ( 1,2). 3. Other autoimmune diseases. Optic neuritis and myelitis are found in systemic lupus erythematosus ( SLE), Sjogren syndrome ( SS), sarcoidosis, vasculitis, Behcet disease, tuberculosis, and paraneoplastic disorders ( 1). WHY IS THE DISTINCTION BETWEEN NEUROMYELITIS OPTICA AND MULTIPLE SCLEROSIS IMPORTANT? The importance of separating NMO from MS is twofold. 1) NMO has a worse outcome than MS, with frequent and early relapses. Within 5 years of onset, 50% of patients are blind in both eyes and cannot walk unassisted, and 20% die of respiratory failure due to cervical myelitis ( 3). 2) NMO responds to immunosuppressive therapy with agents such as azathioprine and rituximab ( 4) and to plasmapheresis, whereas the currently promoted treatment of MS includes immune- modulating agents such as interferon ( 3. THE NEUROMYELITIS OPTICA ANTIBODY Beginning in 2003, Mayo Clinic investigators began publishing articles on NMO that described a serum autoantibody marker and redefined the clinical criteria for NMO diagnosis. In 2004, Lennon et al ( 5,6) identified an IgG marker of NMO that is purported to allow distinction from MS. J Neuro- Ophthalmol, Vol. 27, No. 1, 2007 57 J Neuro- Ophthalmol, Vol. 27, No. 1, 2007 Cross The investigators prospectively tested serum samples from 124 patients with clinical NMO or high risk for NMO. In the first subgroup were 102 North American patients. Of these, 45 had definite NMO, or remained at high risk for NMO and 22 were found to have " classic" MS. NMO was denned as optic neuritis, acute myelitis, and no imaging evidence of demyelinating disease except in the optic nerves and spinal cord. Supportive evidence included either one major criterion or two minor criteria. The major criteria were 1) normal brain MRI at outset, 2) a spinal cord MRI signal abnormality extending three or more vertebral segments, and 3) CSF pleocytosis > 50 X 1( T6 white blood cells/ L or > 5 X 1( T6 neutrophils/ L. The minor criteria were 1) bilateral optic neuritis, 2) severe optic neuritis with irreversible visual acuity loss worse than 20/ 200 in at least one eye, and 3) severe weakness in at least one limb. In the second subgroup were 22 Japanese patients: 11 with definite opticospinal MS, 1 considered to be at high risk of opticospinal MS, 5 with classic MS, and 5 with cerebral infarction. The criteria for the diagnosis of opticospinal MS were exactly those used to diagnose NMO. In addition to the 124 patients with NMO or high risk of NMO, there was a control group of 75 patients with classic MS ( 19 patients), myasthenia gravis ( 10 patients), paraneoplastic visual loss with or without optic neuritis ( 16 patients) Sjogren syndrome ( 12 patients), vasculitis with neurological complications ( 9 patients), and other disorders ( 9 patients) including paraneoplastic myelopathy with CRMP- 5, vitamin B12 deficiency, sarcoidosis, lymphoma, glioma, normal pressure hydrocephalus, and conversion reaction. In addition, further patients were ascertained retrospectively by virtue of incidental detection of an antibody which turned out to be the NMO IgG. These were patients with multifocal neurologic disease suspected of having a paraneoplastic syndrome. Using indirect immunofluorescence on a substrate of mouse central nervous system ( CNS) tissue, the Mayo researchers identified, in the sera of patients with NMO and Japanese opticospinal MS, a distinctive IgG staining pattern localizing to the blood- brain barrier and partly colocalizing with laminin. Seropositivity was present in 60 ( 75%) patients with NMO, 7 ( 58%) Japanese patients with opticospinal MS, and 1 ( 10%) of 10 non- Japanese patients with classic MS, but in none of the North American patients with classic MS, non- MS autoimmune diseases, or other control diseases. Further testing of patients with varying NMO disease burden and activity will be needed to determine the exact incidence and conditions of positivity. THE NEUROMYELITIS OPTICA ANTIBODY AND THE AQUAPORIN- 4 WATER CHANNEL The NMO IgG autoantibody was subsequently shown to bind selectively to the aquaporin- 4 ( AQP4) water channel ( 7), a component of the dystroglycan protein complex located in astrocytic foot processes at the blood-brain barrier. Because of the location of the antigen, it is speculated that this NMO IgG is not merely a marker but a causative agent, as is the case, for example, with the muscle acetylcholine receptor antibodies of myasthenia gravis and the P/ Q- type calcium channel antibodies associated with Lambert- Eaton syndrome. AQP4 is the first water channel-specific autoantibody to be identified. Aquaporin is the predominant water channel in the CNS. Areas of particularly high concentration of this protein are the spinal cord, the optic nerves, the hypothalamus, and the periventricular regions ( 8). The localization of AQP4 in the spinal cord is consistent with the immunopathology of NMO, which affects both gray and white matter, and for unknown reasons is largely restricted to the spinal cord and optic nerves ( 11). IgG, IgM, and products of complement activation are deposited in a perivascular pattern in NMO, suggesting a pathogenic role for the autoantibody ( 9). Blood vessels within demyelinating lesions are thickened and hyalinized. Active lesions exhibit tissue swelling, infiltrating polymorphonuclear macrophages, activated microglia, demyelination, axonal loss, prominent necrosis, and variable degrees of perivascular inflammation with prominence of eosinophils and products of their exocytosis. Chronic lesions show gliosis, cystic degeneration, cavitation, and atrophy. These findings are consistent with a humoral effector mechanism in NMO that is initiated by binding of the NMO autoantibody at the blood- brain barrier ( 9). Pathologically, NMO shares with MS a pattern of focal demyelination, inflammation, scar formation, and axonal destruction but differs in having an intense perivascular response, prominent necrosis, and cavitation ( 9). PREDICTIVE VALUE OF THE NEUROMYELITIS OPTICA ANTIBODY The presence of the NMO IgG autoantibody in the serum of a patient with an initial attack of longitudinally extensive transverse myelitis predicts a myelopathic or optic neuritis relapse within 12 months with more than 50% certainty ( 10). Weinshenker et al ( 10) evaluated 29 patients with a first attack of transverse myelitis with an MRI lesion spanning three or more vertebral segments. Of 23 patients followed for 1 year, 9 were seropositive for the NMO IgG autoantibody. Within 1 year, 5 of 9 patients had a second event, involving recurrent transverse myelitis in 4 patients and optic neuritis in 1 patient. After 1- 7 years of follow- up, none of the 14 patients who were seronegative for the NMO IgG autoantibody had a relapse of myelitis or optic neuritis. In 80 patients with NMO, Wingerchuk et al ( 11) found that mortality due to relapsing NMO was related to a higher attack frequency during the first 2 years of disease and 58 © 2007 Lippincott Williams & Wilkins Neuromyelitis Optica J Neuro- Ophthalmol, Vol. 27, No. 1, 2007 worse motor recovery after the initial myelitis event. These features may facilitate design of treatment trials. Wingerchuk et al ( 12) also studied 96 patients with NMO for the frequency of " secondary progression," as denned by continuous objective deterioration without remission over more than 12 months after one or more attacks. They found that secondary progression was rare in relapsing NMO despite the fact that NMO has a higher attack frequency, and patients tend to be older than those with MS. In contrast to MS, in which attack- free secondary progression accounts for most neurologic impairment, NMO disability was found to be related to clinical relapses ( 12). RECLASSIFYING JAPANESE OPTICOSPINAL MULTIPLE SCLEROSIS AS NEUROMYELITIS OPTICA One of the major points proposed by the Mayo group is that Japanese patients with the opticospinal variant of MS be reclassified as having NMO ( 13,14). In the Japanese opticospinal variant of MS, one does not see a DRB 1* 1501 ( DR2) haplotype association as is seen Western MS. Instead, the DPB 1* 0501 allele has been reported to occur in 90% of the Japanese population with opticospinal MS compared with 60% of Japanese control subjects ( 1). The Japanese patients with opticospinal MS have severe attacks associated with optic neuritis and longitudinally extensive cord lesions, lack oligoclonal bands in the CSF, and have a generally poor prognosis. Nakashima et al ( 15) noted that the IgGl subclass of IgG is not detectable in the CSF of patients with NMO but is elevated in patients with MS. This IgG contributes to the oligoclonal bands seen in MS but not in NMO. Anecdotal reports suggest that interferon ( 3 is not therapeutically effective in opticospinal MS or in NMO but that immunosuppressive drugs may be helpful ( 4). Cree et al ( 2) have recently reported that, compared with Caucasian Americans with MS, African Americans have a greater likelihood of developing opticospinal MS and transverse myelitis and have a more aggressive disease course. It would be interesting to know whether these patients are seropositive for NMO IgG. LUPUS ERYTHEMATOSUS, SJOGREN SYNDROME, AND THE NEUROMYELITIS OPTICA ANTIBODY Pittock et al ( 16) and Weinshenker et al ( 17) have identified an NMO- like disorder in some patients with SLE and SS and in some patients who are antinuclear antibody ( ANA)- positive or extractable nuclear antibody ( ENA)- positive without fulfilling the clinical criteria for SLE or SS. These patients have optic neuritis and extensive spinal cord lesions and are NMO IgG- positive. Pittock et al ( 16) tested serum samples from patients with NMO ( 79), recurrent longitudinally extensive transverse myelitis ( rLETM) ( 44), SLE ( 2), and SS ( 14) for NMO IgG, ANA, and ENA. Approximately three fourths of patients with NMO and rLETM were NMO IgG- positive. Half of these patients were also ANA- positive and about one seventh were ENA-positive. Most did not fulfill the clinical criteria for SLE or SS. There was a higher frequency of nonorgan- specific autoantibodies in NMO IgG- positive patients than in NMO IgG- negative patients. Nonorgan- specific autoantibodies may reflect a more intense autoimmune response. In a similar study, Weinshenker et al ( 17) tested, under masked conditions, the serum of 38 French patients for NMO IgG. Five had uncompheated SS, 5 had uncomplicated SLE, 8 had uncomplicated NMO, 6 had NMO with either SS or SLE, 8 had isolated and/ or recurrent optic neuritis or myelitis with SS or SLE and 6 had SS with neurological disorders other than optic neuritis or myelitis. NMO IgG was detected only in patients with optic neuritis or myelitis. NMO IgG positivity was associated with NMO or NMO partial syndromes in patients with SLE and SS but not with systemic autoimmunity as seen in uncomplicated SLE and SS. MRI IN NEUROMYELITIS OPTICA The issue of brain MRI lesions in NMO has been controversial. Rocca et al ( 18) added magnetization transfer and diffusion tensor sequences to conventional MRI in the examination of 10 patients with NMO and 15 control subjects. On conventional MRI, no macroscopic T2- visible brain lesions were seen in the control subjects or in 6 ( 60%) of the patients with NMO. Two patients with NMO had one T2 lesion each, and 2 had a few nonspecific T2 abnormalities thought to be age- related. But all 10 of the patients with NMO had abnormal proportions of bound and free protons ( decreased magnetization transfer ratio) and increased mean diffusivity within their normal- appearing gray matter, suggesting the presence of tissue damage in areas where conventional Tl and T2 imaging showed no abnormalities. Pittock et al ( 19) addressed the issue of conventional MRI lesions in NMO in a study involving 60 patients with the diagnosis of NMO on the basis of Wingerchuk's 1999 criteria ( See above). Brain MRI lesions were detected in 36 ( 60%). Most were nonspecific, but 6 patients ( 10%) had MS- like lesions, usually asymptomatic. Another 5 patients ( 8%), mostly children, had diencephalic, brainstem, or cerebral MRI lesions atypical for MS. One patient had large cerebral lesions and was comatose. The other patients had subtle lesions. In a large percentage of patients, the MRI brain lesions localized to the sites of high aquaporin protein concentration: the spinal cord, optic nerves, hypothalamus, and periventricular region ( 8). If Pittock et al ( 19) are 59 J Neuro- Ophthalmol, Vol. 27, No. 1, 2007 Cross correct, a substantial percentage of patients with NMO are being misdiagnosed as having MS. REVISING THE DEFINITION OF NEUROMYELITIS OPTICA Based on this work, Mayo Clinic researchers have proposed a revision of the previously published criteria for the diagnosis of NMO ( 3) that had included three absolute requirements: optic neuritis, acute myelitis, and no clinical manifestations implicating other CNS regions. The proposed new criteria ( 20, 21) are optic neuritis, myelitis, and at least two of three supportive features: 1) MRI evidence of a contiguous spinal cord lesion extending over three or more segments at clinical disease onset; 2) brain MRI signal abnormalities not typical for MS, that is, not involving the periventricular region and not scattered in the white matter; and 3) NMO IgG seropositivity. According to these criteria, brain imaging abnormalities found outside the optic nerves are compatible with NMO. Also compatible with a diagnosis of NMO are unilateral optic neuritis alone and optic neuritis and myelitis occurring weeks or even years apart. The Mayo Clinic efforts to characterize NMO have been criticized because they do not fit the original 19th century clinical description of the disorder ( 22). Depending on one's point of view, these studies, taken together, display the asset of internal consistency or the liability of circular reasoning. Critics will need to supply contradictory data. Until that time, the evidence stands in favor of keeping an open mind and considering a new approach to the diagnosis of Devic disease. REFERENCES 1. Weinshenker BG. Neuromyelitis optica: what it is and what it might be. Lancet 2003; 9361: 889- 90. 2. Cree BA, Khan O, Bourdette D, et al. Clinical characteristics of African Americans vs Caucasian Americans with multiple sclerosis. Neurology 2004; 63: 2039^ 5. 3. Wingerchuk DM, Hogancamp WF, O'Brien PC, et al. The clinical course of neuromyelitis optica ( Devic's syndrome). Neurology 1999; 53: 1107- 14. 4. Cree BA, Lamb S, Morgan K, et al. An open label study of the effects of rituximab in neuromyelitis optica. Neurology 2005; 64: 1270- 2. 5. Lennon VA, Lucchinetti CF, Weinshenker BG. Identification of a marker autoantibody of neuromyelitis optica [ abstract]. Neurology 2003; 60( Suppl 1): A519. 6. Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 2004; 364: 2106- 12. 7. Lennon VA, Kryzer TJ, Pittock SJ, et al. IgG marker of optic- spinal multiple sclerosis binds to the aquaporin- 4 water channel. J Exp Med 2005; 202: 473- 7. 8. Pittock SJ, Weinshenker BG, Lucchinetti CF, et al. MRI brain lesions characteristic of neuromyelitis optica ( NMO) colocalize with sites of high aquaporin protein concentrations [ abstract]. Neurology 2006; 66( Suppl 2): A183. 9. Lucchinetti CF, Parisi J, Bruck W The pathology of multiple sclerosis. Neurol Clin 2005; 23: 77- 105. 10. Weinshenker BG, Wingerchuk DM, Vukusic S, et al. Neuromyelitis optica IgG predicts relapse after longitudinally extensive transverse myelitis. Ann Neurol 2006; 59: 566- 9. 11. Wingerchuk DM, Weinshenker BG. Neuromyelitis optica: clinical predictors of a relapsing course and survival. Neurology 2003; 60: 848- 53. 12. Wingerchuk D, Pittock S, Lennon V, et al. Secondary clinical progression is rare in relapsing neuromyelitis optica [ abstract]. Neurology 2006( Suppl 2): A380. 13. Weinshenker B. Western vs optic- spinal MS: two diseases, one treatment? Neurology 2005; 64: 594- 5. 14. Weinshenker BG, Wingerchuk DM, Nakashima I, et al. OSMS is NMO, but not MS: proven clinically and pathologically. Lancet Neurol 2006; 5: 110- 1. 15. Nakashima I, Fujihara K, Fujimori J, et al. Absence of IgGl response in the cerebrospinal fluid of relapsing neuromyelitis optica. Neurology 2004; 62: 144- 6. 16. Pittock SJ, Lennon VA, Wingerchuk DM, et al. The prevalence of non- organ- specific autoantibodies and NMO- IgG in neuromyelitis optica ( NMO) and related disorders [ abstract]. Neurology 2006; 66( Suppl 2): A307. 17. Weinshenker B, De Seze J, Vermersch P, et al. The relationship between neuromyelitis optica and systemic autoimmune disease [ abstract]. Neurology 2006; 66( Suppl 2): A380. 18. Rocca MA, Agosta F, Mezzapesa DM, et al. Magnetization transfer and diffusion tensor MRI show gray matter damage in neuromyelitis optica. Neurology 2004; 62: 476- 8. 19. Pittock SJ, Lennon VA, Krecke K, et al. Brain abnormalities in neuromyelitis optica. Arch Neurol 2006; 63: 390- 6. 20. Wingerchuk DM, Pittock SJ, Lennon VA, et al. Neuromyelitis optica diagnostic criteria revisited: validation and incorporation of the NMO- IgG serum autoantibody [ abstract]. Neurology 2005; 64( Suppl 1): A38. 21. Wingerchuk DG, Lennon VA, Pittock SJ, et al. Revised diagnostic criteria for neuromyelitis optica. Neurology 2006; 66: 1485- 9. 22. Devic E. Myelite aigue dorse- lombaire avec nevrite Optique, autopsie. Vol. 1. Congres Francais Med. Lyon, Premiere Session; 1895: 434- 9. 60 © 2007 Lippincott Williams & Wilkins |