| Title |
Influence of Pyrites on Furnace Slagging |
| Creator |
Bryers, Richard W.; Walchuk, O. R. |
| Publisher |
University of Utah |
| Date |
1983 |
| Spatial Coverage |
Akron, Ohio |
| Abstract |
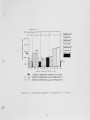
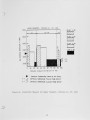
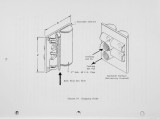
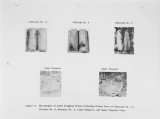
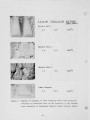
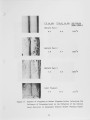
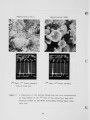
Traditional, analytical techniques used to evaluate the quality of coal are not always successful in predicting a fouling or slagging condition. On occasions different coals with identical ash chemistry have produced decidedly different slagging conditions in boilers of identical design operated in the same mode. Variations in composition of the slag when compared to coal ash suggested specific minerals are being selectively deposited on furnace walls depending upon their specific gravity, composition, and physicochemical properties. Five coals have been analyzed using size and gravity fractionation techniques as applied to pulverized coal to determinethe relative mineral distribution in the coal. These coals have been fired in a 100 lb/h laboratory furnace to determine the impact of mineral distribution on slagging. This paper describes the results. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s6pg1v8v |
| Setname |
uu_afrc |
| ID |
53 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s6pg1v8v |