| Title |
Heat Recovery From Oxy-Fuel Based Glass Melting Processes |
| Creator |
Parekh, Uday N.; Chen, Michael S. K.; Painter, Corning F. |
| Publisher |
University of Utah |
| Date |
1990 |
| Spatial Coverage |
presented at San Francisco, California |
| Abstract |
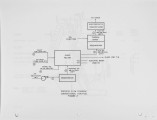
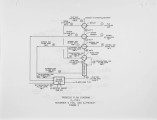
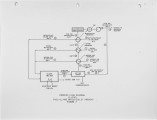
In a development program, supported by the U.S. Department of Energy and involving close interaction with the glass industry, Air Products and Chemicals, Inc. has proposed three new oxygen based Thermally Efficient Alternative Melter (TEAM) processes that aim to reduce emissions and help improve the competitiveness of the U.S. glass industry. These processes are lower in capital costs, as well as more energy efficient and environmentally benign compared to current production methods. Use of oxygen in combustion results in significantly lower sensible heat loss through the stack and higher melter thermal efficiencies. This inherent advantage of an oxy·-fuel process is augmented further by the heat recovery and integration schemes that are described in this paper. The need for costly regenerators, conventionally used in glass manufacture, is eliminated. Also, the reduction in particulates and NOx and SOx emissions resulting from an oxy-fuel conversion eliminates the need for expensive pollution control equipment and cuts the use of costly electric boosting. The three heat recovery schemes investigated to further improve the intrinsic economics of oxy-fue1 based glass melting are: 1) raw material preheating, 2) gas turbine cycles, and 3) natural gas reforming. These TEAM process schemes can improve energy efficiency by up to 24% and provide capital savings of up to 50% over the conventional air based process. These potential savings, along with the important environmental benefits associated with oxygen usage have generated considerable enthusiasm from the glass industry. Key equipment from one of the TEAM processes will be designed and demonstrated in partnership with a glass manufacturer in the next phases of this development program. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s67s7rc9 |
| Setname |
uu_afrc |
| ID |
6205 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s67s7rc9 |