| Title |
State-of-the-Art Ultra Low NOx Burner |
| Creator |
Nutcher, Peter B.; Lewandowski, David A.; Waldern, Peter J. |
| Publisher |
University of Utah |
| Date |
1992 |
| Spatial Coverage |
presented at Cambridge, Massachusetts |
| Abstract |
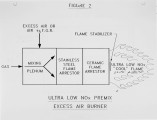
Nitrogen Oxide (NOx) emission limits from combustion sources are becoming increasingly stringent. For example, the Southern California Air Quality Management District (SCAQMD) requires new sources to comply with Best Available Control Technology (BACT). Currentiy, the SCAQMD considers BACT to be about 20 ppmv (at 3% 02) and even this limit is expected to decrease in the future. Rules of this type are expected to proliferate as regulations stemming from the 1990 Clean Air Act Amendments are promulgated. Process Combustion Corporation has developed an ultra low NOx burner which can meet all existing NOx regulations with ample margin. In fact, the burner has consistently achieved NOx emissions below 10 ppmv (dry, corrected to 3% 02) or 0.012Ib/MM BTU in laboratory tests. These same tests have demonstrated excellent flame stability and CO levels also below 10 ppmv. The burner prototype consists of a basic 5 MM BTU/hr module. For commercial applications, multiple burner modules will be supplied to meet larger process requirements. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s65d8vfd |
| Setname |
uu_afrc |
| ID |
11597 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s65d8vfd |