| Title |
Slagging Behavior of Coal Ash in an Experimental Furnace |
| Creator |
Singh, S. N.; Austin, L. G.; Krauss, W. E.; Essenhigh, R. H. |
| Publisher |
University of Utah |
| Date |
1982 |
| Spatial Coverage |
presented at Newport Beach, California |
| Abstract |
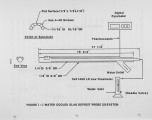
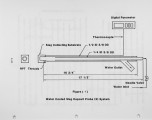
Coal has been fired in a laboratory-scale plug flow furnace to study slag deposition on water-cooled slagging tubes at the bottom of the furnace. The problem of slagging is a major factor of boiler availability; it has been under investigation for decades but it is still regarded as being only partly controllable where "control" has often been possible only by de-rating. Slagging mostly occurs at the level of the highest flame temperatures, but it can occur on the upper furnace walls; it can be particularly disruptive if it occurs on the superheater tubes. Boilers have sometimes been observed to remain clean for substantial periods of time; but when slagging does occur, it can do so with great rapidity. This has been interpreted as a process of surface "conditioning." Once adhesion starts, even in a small patch, the thermal insulation effect allows the gas side surface of the patch to become even more adhesive because of the rise in surface temperature. If the patch surface slags, molten slag can flow down to cover and adhere to bare tube areas below. The patch also provides a location that may accelerate local "conditioning" and stimulate growth. With large enough deposits there can be substantial imbalance of heat transfer to the water. Locally, if the tube side of the deposit gets cold enough it can then freeze close to the tubes, and on occasion will crack off. Slag masses falling from the upper furnace have severely damaged or even broken the tubes above the ash pit. |
| Type |
Text |
| Format |
application/pdf |
| Language |
eng |
| Rights |
This material may be protected by copyright. Permission required for use in any form. For further information please contact the American Flame Research Committee. |
| Conversion Specifications |
Original scanned with Canon EOS-1Ds Mark II, 16.7 megapixel digital camera and saved as 400 ppi uncompressed TIFF, 16 bit depth. |
| Scanning Technician |
Cliodhna Davis |
| ARK |
ark:/87278/s6222x9s |
| Setname |
uu_afrc |
| ID |
3232 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s6222x9s |