| OCR Text |
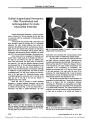
Show ORIGINAL CONTRIBUTION Neuro- Ophthalmologic Features of Spinocerebellar Ataxia Type 7 Randee C. Miller, BS, Aash Tewari, MD, Joel A. Miller, MD, James Garbern, MD, and Gregory P. Van Stavern, MD Background: Spinocerebellar ataxia type 7 ( SCA7) is a progressive ataxia that is unique among inherited ataxias in having a high prevalence of retinal photo-receptor abnormalities. However, the ophthalmic fea-tures and their relationship to the neurologic features of SCA7 have not been widely reported. The goal of this study was to provide increased documentation. Methods: The medical records of 10 consecutive patients with SCA7 examined in the Neuro- Ophthal-mology Clinic at Kresge Eye Institute between 2000 and 2008 were reviewed retrospectively. Each patient underwent a standardized ophthalmologic and neu-rologic examination. Some patients also underwent electroretinography ( ERG). Eight patients had ge-netically confirmed disease and 2 patients had presumptive SCA7 based on their clinical pre-sentation. Patients were excluded if they had visual loss or ataxia due to other causes. Results: Nine patients reported visual symptoms at presentation, including hemeralopia, photophobia, dyschromatopsia, and blurred vision. In 3 of these patients, the visual symptoms had preceded the onset of ataxic symptoms. Visual acuity was abnormal in all patients at presentation. Four patients with visual dysfunction had normal or minimally abnormal macular pigmentary changes, but all patients had abnormal electroretinograms ( ERGs) showing pri-marily cone dysfunction. The severity of visual loss and the severity of ataxia were frequently discordant. Conclusions: Based on this study, patients with SCA7 often have visual symptoms that may precede, accompany, or follow the onset of ataxic symp-toms. The severity of vision loss and ataxia may be discordant. Ophthalmoscopic evidence of macular abnormalities may be scant, but results of ERG will always be abnormal. This information may assist in earlier and more cost- effective diagnosis and permit more effective patient counseling. ( J Neuro- Ophthalmol 2009; 29: 180- 186) S pinocerebellar ataxia type 7 ( SCA7) is a rare autosomal dominant, slowly progressive neurodegenerative disor-der associated with abnormal expansion of a CAG trinu-cleotide repeat within the ATXN7 gene on chromosome 3p12- 13 ( 1). Normal alleles have less than 20 CAG repeats; approximately 75% of normal alleles have 10 repeats. Disease- causing alleles have greater than 36 CAG repeats ( 1). SCA7 is the only inherited ataxia invariably associated with a cone- rod dystrophy ( 1,2). Patients with cone- rod dystrophy present with reduced visual acuity, dyschromatopsia, and photosensitivity, followed by pro-gressive loss in peripheral vision and night blindness ( 2). On ophthalmoscopic evaluation, such patients may initially have a normal- appearing macula. Eventually retinal pig-ment epithelial mottling occurs in the macular area, finally developing into a ‘‘ bull's- eye'' maculopathy. Full- field electroretinography ( ERG) usually shows loss of photopic function, reflecting loss of cone photoreceptors. In the early stages of the disease, multifocal ERG may be more sensitive than full- field ERG. Slow saccadic eye movements are common but not specific for SCA7. The cerebellar ataxia manifests first as gait imbalance, later joined by appendicular ataxia, and eventually leading to wheelchair confinement. Other neurologic findings in SCA7 include hyperreflexia, spasticity, dysarthria, and dysphagia ( 3). SCA7 exhibits genetic anticipation caused by expan-sion of the CAG repeats in offspring of affected males. The anticipation applies to the age of onset and rate of progression of disease, so that affected offspring experience symptoms at an earlier age and the disease progresses at a much faster rate than in the affected parent. A patient may become symptomatic before the transmitting parent, and Departments of Ophthalmology ( RCM, JT, JAM, GPV) and Neurology ( JG, GPV) Wayne State University, Detroit, Michigan. This study was supported by an unrestricted grant from Research to Prevent Blindness to the Department of Ophthalmology at Wayne State University. Address correspondence to Gregory P. Van Stavern, MD, Department of Ophthalmology and Visual Sciences, Washington University in St. Louis, 660 South Euclid Avenue, St. Louis, MO 63110; E- mail: vanstaverng@ vision. wustl. edu 180 J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 thus there may be an apparent absence of a family history of the disorder ( 1). The clinical course of SCA7 is variable with regard to age of onset and rate of progression. Patients may present with symptoms of ataxia, symptoms of visual loss, or both ( 3,4). The initial manifestations of SCA7 may be subtle, and affected patients may be misdiagnosed until their symptoms are advanced, resulting in unnecessary testing, incorrect treatment, and inaccurate prognosis. Previous studies of SCA7 are limited and lack detail regarding the visual manifestations ( including macular changes) and how these manifestations correlate with the neurologic features. In evaluating 26 patients with SCA7, Martin et al ( 3) provided detailed information regarding the number of triplet repeats and electrophysiologic data but did not report visual acuity or detailed visual and neuro-logic examination features. In a study of only 3 patients with SCA7, Ahn et al ( 4) formally evaluated visual function, fundus findings, and visual electrophysiology but did not evaluation neurologic function and the relationship between the two sets of symptoms. Our study was designed to provide more information on the neuro- ophthalmologic manifestations of SCA7, particularly their relationship to neurologic dysfunction. METHODS The medical records of 10 consecutive patients with SCA7 seen in the Neuro- Ophthalmology Clinic of the Kresge Eye Institute, Wayne State University, Detroit, Michigan, between 2000 and 2008 were retrospectively reviewed. All patients were referred for unexplained visual loss, and none of the patients had a specific genetic diag-nosis. Most of the patients had had a recent ophthalmologic examination without an explanation for complaints of visual loss. All patients underwent a standardized evaluation at time of presentation, including measurement of visual acuity, color vision, and ocular motility, together with dilated ophthalmoscopy and a full neurologic examination. Ancillary tests, including perimetry, full- field ERG, and optical coherence tomography ( OCT), were performed in some patients. Eight patients ultimately had genetically confirmed disease; 2 patients had presumptive SCA7 based on clinical presentation and a suggestive family history. Patients who returned for follow- up examinations were re- evaluated at each follow- up appointment. Patients were excluded if they had visual loss or ataxia due to other causes. We determined the age of disease onset, initial symptoms, and duration of illness based on the patient's recollection. All patients with noncoincident visual and neurologic symptoms were asked to specify the estimated gap. The best they could offer was ‘‘ several years,'' and we could not verify the accuracy of this information. All patients were assigned a disease stage corre-sponding to their degree of disability at presentation. Dis-ease stages were defined as follows: stage 0, no gait difficulties; stage 1, self- reported difficulty with gait; stage 2, loss of independent gait as defined by permanent use of a walking aid or reliance on a supporting arm; and stage 3, confinement to a wheelchair ( 5). Color vision testing was assessed using Ishihara color plates. Patients who were unable to identify the control plate were not included in color vision analysis. Snellen visual acuity was obtained using best optical correction. Macular changes were graded based on the ophthalmo-scopic examination by the neuro- ophthalmologist ( GPV). Family history was considered positive if there was at least one first- degree relative with ataxia, not necessarily confirmed genetically as SCA7. RESULTS The initial symptoms experienced by patients in-cluded visual dysfunction, neurologic dysfunction, or a combination of the two. Nine patients had visual symptoms at presentation, either alone or in combination with neuro-logic symptoms ( Table 1). Three of 10 patients reported visual loss before the onset of ataxia, 1 reported ataxia before the onset of visual loss, 5 reported simultaneous onset of visual and neurologic symptoms, and 1 had unknown order of onset. Visual manifestations included hemeralopia ( 7 patients), photo-phobia ( 4 patients), dyschromatopsia ( 10 patients), and blurred vision ( 2 patients). At presentation, all patients had measurable visual loss ( average visual acuity of 20/ 250 right eye and 20/ 200 left eye) and dyschromatopsia ( average number of correctly identified Ishihara color plates: 25% right eye and 27% left eye). Macular changes ranged from mild granular retinal pigment epithelium changes to a bull's- eye maculopathy in more severely affected patients ( Fig. 1). Four patients had either normal- appearing maculae or minimal macular changes, despite reduced visual acuity and evidence of cone dysfunction on ERG. The 8 patients with genetically confirmed SCA7 had a family history of visual loss and ataxia, although not always initially recognized as manifestations of SCA7. The 2 patients with suspected SCA7 had a family history of ataxia but no definite visual loss. Most patients required assistance for ambulation. All patients had full versions but with saccadic smooth pursuit, slow saccades, and saccadic dysmetria. Convergence was normal, although this was not formally 181 Neuro- Ophthalmic Features of SCA7 J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 TABLE 1. Clinical features of our cohort of 10 patients with SCA7 Case Race, Gender Age at Presentation* ( years) Age at Onset** ( years) Time to Presentation† ( years) Initial Symptom Snellen Visual Acuity ( OD, OS) Ophthalmoscopic Macular Changes Ataxia Symptoms at Presentation Ataxia Stage‡ CAG Repeats Family History 1 AA, M 46 41 5 Gait ataxia 20/ 70, 20/ 50 Moderate 1 1 45 1 2 AA, M 61 12 49 Visual loss 20/ 400 OU Severe 1 2 46 1 3 AA, F 59 44 15 Both 20/ 200 OU Severe 1 3 45 1 4 AA, F 58 48 10 Both 20/ 50 OU Moderate 1 1 39 1 5 AA, M 5 4 0.5 Both 1/ 200 OU Moderate 1 1 Unknown 1 6 AA, F 37 36 0.67 Both 20/ 100, 20/ 200 None 1 1 45 1 7 ME, M 47 43 4 Visual loss 20/ 200, 20/ 100 Mild 0 44 1 8 AA, F 44 35 9 Both 1/ 200, 3/ 200 Severe 1 2 45 1 9 AA, M 48 40 8 Visual loss 20/ 200 OU Mild 1 3 Unknown 10 ME, F 45 43 1.5 Gait ataxia 20/ 40, 20/ 30 Mild 1 1 Unknown Mean 45 34.6 10.3 20/ 250, 20/ 200 44 * The age at which a patient presented to the neuro- ophthalmologist. ** The age at which the patient first experienced symptoms. In patients who had a delay between the onset of visual and neurologic symptoms, the delay time was measured in years, but these patients were unable to provide a more specific length of time. † Time delay from symptom onset to presentation to physician. ‡ Refers to degree of ataxia as follows: Stage 0, no gait difficulties; Stage 1, disease onset, with gait ataxia; Stage 2, loss of independent gait; and Stage 3, confinement to a wheelchair. AA, African American; M, male; ME, Middle Eastern; OD, right eye; OS, left eye; OU, both eyes; W, female. 182 q 2009 Lippincott Williams & Wilkins J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 Miller et al evaluated in all patients with near point of convergence and convergence amplitudes. None of the patients had nystagmus in primary gaze, but all had horizontal eccentric gaze jerk nystagmus in the direction of gaze. Visual fields were performed in 8 patients ( Hum-phrey automated perimetry in 7 and Goldmann kinetic perimetry in 1). One patient had a normal Humphrey visual field test, 3 had moderate generalized depression, and 4 had central scotomas ( including the patient who underwent Goldmann perimetry). Results of full- field ERG, performed in 4 patients, were always abnormal, showing reduced cone responses in 4 patients and reduced cone and rod responses in 1 patient. At the last follow- up visit, all 10 patients had a wide-based ataxic gait with impaired balance and coordination and varying degrees of disability. Four patients needed ambulation assistance ( stage 2), and two were wheelchair-bound ( stage 3). The number of abnormal CAG repeats in patients who received testing ranged from 39 to 46; the age of onset FIG. 1. Case 6. A. Fundus photographs performed 2 years after our initial examination show subtle granular retinal pigment epithelial changes. B. Three years later, these changes are much more evident and mild temporal optic disc pallor has developed. 183 Neuro- Ophthalmic Features of SCA7 J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 TABLE 2. Previously published studies and this study of ophthalmic and neurologic features of SCA7 Study No. of Patients No. of Families Visual Function Neurologic Function Conclusions Martin et al ( 3) 26 1 Evaluated with ERG, visual evoked potentials, and ophthalmologic examination; visual acuities not formally reported Evaluated with clinical neurologic examination and brainstem auditory evoked potentials Inverse correlation found between number of CAG repeats and age at onset of disease Michalik et al ( 6) 212 NA Ophthalmologic features of SCA7 summarized based on review of previously published studies Neurologic features of SCA7 summarized based on review of previously published studies Review and summary of clinical, pathologic, and genetic features of SCA7 from previously published studies Aleman et al ( 8) 3 1 Evaluated with ophthalmologic examination, Goldmann kinetic perimetry, dark- adapted and light- adapted static threshold perimetry, full field and focal ERG, and OCT Not formally assessed Retinopathy seen in SCA7 found to have the phenotype of a cone- rod dystrophy; spectrum of severity ranged from mild to severe dysfunction Gu et al ( 7) 8 2 Evaluated with ophthalmic examination including visual acuity, color discrimination, and direct ophthalmoscopy Clinical neurologic examination Of two families studied, there was marked variation in age at onset, initial symptoms, and disease; all patients experienced visual loss and ataxia Ahn et al ( 4) 3 1 Evaluated with funduscopic examination, full- field ERG, electro- oculography, and OCT; macular functions assessed with mfERG and scanning laser ophthalmoscopy microperimetry Not formally assessed Macular functions severely depressed in the ophthalmoscopically visible atrophic areas and in the areas without visible atrophy in a centrifugal pattern; areas of functional deficits were greater than the areas of anatomic deficits Hugosson et al ( 9) 3 2 Evaluated with ophthalmologic examination including visual acuity, fundus inspection, Farnsworth color vision test, Goldmann perimetry, full- field ERG, mfERG, and OCT Not formally assessed Patients presented with early macular dysfunction before any ophthalmoscopically visible abnormalities were noted; Based on the ERG findings, the primary dysfunction involved the cone photoreceptors in the foveal region ( Continued on next page) 184 q 2009 Lippincott Williams & Wilkins J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 Miller et al in this group ranged from the 2nd to the 5th decade. These results are similar to those of other published studies ( 1,6). DISCUSSION Our study highlights many of the key features of SCA7, including the differences in severity and time of onset of the visual loss and the ataxia, the lack of obvious visible macular changes early in the disease, the potential for misdiagnosis, and the relationship between age of onset and number of CAG repeats. Previous studies of SCA7 ( Table 2) have not documented the degree of visual loss in relation to neurologic findings and have underemphasized the importance of identifying cone dystrophy. Compared with previous reports, our study includes a relatively large number of families. Table 1 reveals that the severity of visual loss and the severity of ataxia were often discordant. Although 5 patients reported that visual and ataxic symptoms began at the same time, 3 experienced visual symptoms several years before the onset of ataxic symptoms, and 1 had ataxic symptoms for several years before experiencing visual loss. We acknowledge that our information about timing of symptom onset was based on self- recollection. Even so, we are certain that visual loss preceded ataxia by several years in 3 patients, a reminder that SCA7 may present initially with isolated cone dysfunction. None of the patients had been genetically identified as having SCA7 when we first examined them, either for their visual or their neurologic symptoms. Some of the patients were being followed by a neurologist and an ophthalmologist, neither of whom had considered a unify-ing diagnosis, perhaps because the patients did not offer their visual symptoms of photosensitivity and hemeralopia without our probing. As we noted, such symptoms can be present even when the macular changes are minimal and visual loss is mild, but full- field ERG will always identify features of cone dysfunction. Early diagnosis of SCA7 might assist patient counseling regarding disease prognosis, aid in the identification of asymptomatic family members, and result in more cost- effective diagnostic testing. A complete hereditary ataxia panel from Athena Diagnostics ( www. athenadiagnostics. com) costs more than $ 12,000, whereas testing specifically for SCA7 costs approximately $ 650. Although no specific treatment is available, a correct diagnosis may prevent unnecessary diagnostic testing, allow patients access to multidisciplinary visual and neurologic rehabilitation services, and secure the diagnosis of affected family members. Indeed, the mother of one of our affected patients had a diagnosis of multiple sclerosis. Once her daughter received a confirmed diagnosis, she was reassessed by her neurologist and found to have SCA7 TABLE 2. Continued Study No. of Patients No. of Families Visual Function Neurologic Function Conclusions Maschke et al ( 10) 7 2 Not formally assessed Clinical neurologic examination SCA7 was associated with upper motor neuron signs, reduced eye movement velocity, reduced cough, and head- eye lag Miller et al ( this study) 10 9 Evaluated with standardized ophthalmologic examination including visual acuity, color discrimination, and dilated ophthalmoscopic examination; some patients also underwent perimetry testing, ERG, and OCT Clinical neurologic examination, including assessment of ocular motility and cerebellar functions Patients with SCA7 presented with visual loss, ataxia, or both; severity of ataxia and visual loss were often discordant ERG, electroretinogram; mfERG, multifocal ERG; NA, not applicable; OCT, optical coherence tomography; SCA7, spinocerebellar ataxia type 7. 185 Neuro- Ophthalmic Features of SCA7 J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 rather than MS and treatment for MS was discontinued. Several patients in this series were referred to a multidis-ciplinary visual rehabilitation service with benefit, empha-sizing that the lack of specific treatment does not negate the importance of supportive and rehabilitative therapies. SCA7 should be a diagnostic consideration when a patient presents with ataxia and evidence of a cone- rod dystrophy, especially in the setting of a positive family history. However, the absence of a family history or of ophthalmoscopically evident macular changes does not preclude the diagnosis. A strong clinical suspicion of SCA7 should prompt selective molecular genetic testing or referral to specialist familiar with such conditions. REFERENCES 1. Bird TD, Pagon RA, La Spada AR. Spinocerebellar ataxia type 7. NCBI GeneReviews Web site. http:// www. ncbi. nlm. nih. gov/ bookshelf/ br. fcgi? book= gene& part= sca7. Accessed June 1, 2009. 2. Hamel C. Cone rod dystrophies. Orphanet J Rare Dis 2007; 2: 7. 3. Martin J- J, Van Regemorter N, Del- Favero J, et al. Spinocerebellar ataxia type 7 ( SCA7)- correlations between phenotype and genotype in one large Belgian family. J Neurol Sci 1999; 168: 37- 46. 4. Ahn JK, Seo JM, Chung H, et al. Anatomical and functional characteristics in atrophic maculopathy associated with spinocer-ebellar ataxia type 7. Am J Ophthalmol 2005; 139: 923- 5. 5. Schmitz- Hu ¨ bsch T, du Montcel ST, Baliko L, et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology 2006; 66: 1717- 20. 6. Michalik A, Martin JJ, Van Broeckhoven C. Spinocerebellar ataxia type 7 associated with pigmentary retinal dystrophy. Eur J Hum Genet 2004; 12: 2- 15. 7. Gu W, Wang Y, Liu X, et al. Molecular and clinical study of spinocerebellar ataxia type 7 in Chinese kindreds. Arch Neurol 2000; 57: 1513- 8. 8. Aleman TS, Cideciyan AV, Volpe NJ, et al. Spinocerebellar ataxia type 7 ( SCA7) shows a cone- rod dystrophy phenotype. Exp Eye Res 2002; 74: 737- 45. 9. Hugosson T, Gra ¨ nse L, Ponjavic V, et al. Macular dysfunction and morphology in spinocerebellar ataxia type 7 ( SCA7). Ophthalmic Genet 2009; 30: 1- 6. 10. Maschke M, Oehlert G, Xie TD, et al. Clinical feature profile of spinocerebellar ataxia type 1- 8 predicts genetically defined subtypes. Mov Disord 2005; 20: 1405- 12. 186 q 2009 Lippincott Williams & Wilkins J Neuro- Ophthalmol, Vol. 29, No. 3, 2009 Miller et al |