| OCR Text |
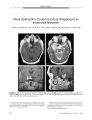
Show ORIGINAL CONTRIBUTION Bilateral Occipital Lobe Hyperperfusion Demonstrated by Single Photon Emission Computed Tomography During Seizure- Related Cortical Blindness Jorge C. Kattah, MD, Meena Gujrati, MD, Edward T. Hui, MD, and Charles C. Chen, MD Abstract: An 81- year- old woman with chronic dementia developed lethargy, confusion, binocular blindness, and episodic left- beating nystagmus. Diffusion magnetic resonance imaging ( MRI) revealed high signal in the right occipital region suggesting recent ischemia. A concurrent electroencephalogram ( EEG) showed a right occipital seizure focus that spread to the opposite occipital lobe. A single photon emission computed tomography ( SPECT) performed during the seizure epoch showed bilateral occipital lobe hyperperfusion. This is the second report to document SPECT bi- occipital hyperperfusion in seizure-related cortical blindness. ( JNeuro- Ophthalmol 2004; 24: 24- 26) Cortical blindness is an infrequent manifestation of occipital lobe epilepsy ( 1- 4). To explain bilateral visual loss on the basis of a unilateral occipital seizure focus, one must infer spread of the seizure across the splenium of the corpus callosum to disable the contralateral occipital cortex. We report clinical, electroencephalographic ( EEG), and single photon emission computed tomography ( SPECT) evidence that supports this hypothesis. A previous report ( 9) has described one case of bilateral occipital hyperperfusion on ictal SPECT in post- ictal blindness. CASE REPORT In 1999, an 81- year- old woman suddenly developed confusion and intermittent visual hallucinations characterized by red, purple, and white lights lasting a few minutes. Within hours, she developed leftward deviation of the eyes, bursts of left- beating nystagmus, and decreased vision. She was confused, lethargic, and complaining of blindness. From the Departments of Neurology ( JCK, MG, ETH) and Pathology ( MG), and the Division of Neuro- pathology and Radiology ( CCC), University of Illinois College of Medicine, Peoria, and the Illinois Neurologic Institute at OSF Saint Francis Medical Center, Peoria, Illinois. Address correspondence to Jorge C. Kattah, MD, Department of Neurology, University of Illinois College of Medicine at Peoria located at OSF Saint Francis Medical Center, 530 N. E. Glen Oak Ave., Peoria, IL 61637. E- mail Address: kattahj@ uic. edu Four years earlier, she had begun to develop recent memory loss, behavioral and personality changes, and a progressive cognitive deficit that elicited a diagnosis of probable Alzheimer's disease. Six months earlier, she had had a Mini- mental State scale score of 24/ 30 and was able to perform activities of daily living with minimal assistance. The last recorded routine ophthalmologic evaluation in 1997 had disclosed best- corrected visual acuities of 20/ 30 OU, normal confrontation visual fields, and normal eye movements. Our initial examination, on the day after the onset of her acute blindness, showed a patient who followed simple commands. Although she failed to identify objects visually, she could name them correctly by sound or touch. She displayed 40- to 60- second bursts of left- beating nystagmus and left gaze deviation without head deviation, during which she had no light perception. Between nystagmus episodes, the eyes were positioned in midline and she had light perception. She was pseudophakic and the pupils were of normal shape and size and reacted briskly to light. The ophthalmoscopic examination was normal. Optokinetic nystagmus ( OKN) was absent. An electroencephalogram ( EEG) performed immediately before onset of a nystagmus episode demonstrated right occipital lobe spike discharges with a frequency of 14 to 18 Hz ( Fig. 1 A). The EEG tracing after nystagmus onset revealed seizure spread to the left occipital lobe ( Fig. IB). During non- nystagmus periods, the EEG showed generalized delta slowing, intermixed with 2 Hz spike- and- slow wave discharges from the right occipital lobe. Two doses of 250 mg of phenytoin and 1 mg of lorazepam were administered intravenously over a 30- minute period, but the clinical manifestations did not change. The patient underwent brain magnetic resonance imaging ( MRI), which showed no abnormalities on T2- weighted sequences, but there was an area of increased signal intensity on the diffusion- weighted image ( DWI) in the right medial occipital region suggestive of ischemia ( Fig. 2). A SPECT scan, performed 30 minutes after the MRI and during a period in which the patient was blind and had 24 J Neuro- Ophthalmol, Vol. 24, No. 1, 2004 Cortical Blindness and Ictal Nystagmus JNeuro- Ophthalmol, Vol 24, No. 1, 2004 ^ fr^* W\ if^ f* W^<$#^^) w\ t^ *, « / yyH^>^ YV^ A* riA » * ( V^ v^^ « / vuvVy ^ wv^ M^ Av^ v^ v^ f^ W- vy,,**>' t/ jv'**' vi#'' y4* it> nystagmus FIGURE 1. A. Electroencephalogram performed before the onset of nystagmus. The epileptic discharge originates from the right occipital lobe ( P4- 02, T6- 02) with an average spike frequency of 14 Hz. B. Electroencephalogram obtained at the onset of nystagmus ( recorded with AC current electro- oculogram electrodes LEOG, REOG). The discharge has spread to the right fronto- temporal ( F4- C4, F8- T4) and left occipital ( T5- 01, P3- 01) regions. episodes of left- beating nystagmus, demonstrated bilateral and symmetric areas of increased cerebral blood flow in the striate cortex and the splenium in comparison with the normal blood flow in the cerebellum ( Fig. 3). Over the next 24 hours, the patient received several intravenous doses of phenytoin, phenobarbital, and loraze-pam. A follow- up EEG showed generalized slowing in the theta range with resolution of the partial occipital lobe seizures. The episodes of nystagmus disappeared and visual acuity improved to 20/ 400 OU. However, the patient could follow only simple commands. One month later, her mini mental status scale score was 6/ 30. She had regained a visual acuity of 20/ 80 in the OD and 20/ 400 in the OS. She could read words but no sentences; she identified objects, colors, and familiar faces. Confrontation visual fields were normal. The pupil, ophthalmoscopic, and oculomotor examinations were normal, although the OKN response remained absent. An EEG showed no seizure activity. Five weeks later, the patient became quadriplegic and died. On autopsy, massive infarction of the pons secondary to acute thrombosis of the right vertebral and distal basilar arteries was identified as the cause of death. The brain also showed diffuse gyral atrophy with frontoparietal predominance. The microscopic examination revealed changes of Alzheimer's disease and beta amyloid deposition in the small arteries and arterioles of the cortex and leptome-ninges. The amyloid angiopathy was most prominent in the occipital lobes, the latter also showing reactive gliosis, especially around thick walled vessels. The region of the seizure focus showed extensive gliosis. There were lacunar infarcts in the left caudate and right putamen and diffuse small vessel disease of the white matter of moderate severity. Sections of the occipital lobe with attention to the presumed location of the seizure focus did not show infarction. DISCUSSION Our patient demonstrated cortical blindness during a period of frequent partial occipital seizures believed to be triggered by a small acute right occipital stroke. Once her seizures were arrested with anti- convulsants, she regained FIGURE 2. Axial MRI. The T2 sequence ( left) shows no abnormalities. The diffusion image ( right) shows an area of hyperintensity in the right medial occipital lobe that may represent ischemia. 25 JNeuro- Ophthalmol, Vol. 24, No. 1, 2004 Kattah et al. nn [ jStsJ ua nono E^ JL^ t^ LJ UDDD FIGURE 3. Single photon emission computed tomography ( SPECT) of the brain performed during nystagmus episode and blindness ( top row = axial plane, middle row = sagittal plane, bottom row = coronal plane). It shows relatively increased blood flow ( white) in the occipital cortices and the corpus callosum relative to the normal blood flow ( yellow) in the cerebellum. Relatively reduced blood flow ( pink) in the temporal and parietal lobes is a feature of Alzheimer's disease. sight. SPECT documented bi- occipital hyperperfusion during the epoch of her frequent seizures. Cortical blindness occurring in occipital lobe epilepsy now has been recognized for more than 120 years ( 1). Although Penfield and Jasper ( 2) considered it a frequent manifestation in this setting, cortical blindness was not reported in a retrospective study of 55 patients with occipital lobe epilepsy ( 3). On the other hand, another study identified 10 patients with ictal amaurosis among 25 patients with occipital lobe epilepsy ( 4). The distinctive feature of our report is the finding of SPECT hyperperfusion of both occipital lobes during the period of frequent seizures. In a previous report, ictal SPECT in six patients with occipital lobe epilepsy without ictal cortical blindness or nystagmus showed ipsilateral occipital and temporal lobe hyperperfusion in three patients and bilateral occipital temporal and parietal lobe hyperperfusion in three patients ( 8). In a patient who reported a 15- minute period of post- ictal cortical blindness, and whose EEG showed focal motor seizures and occipital lobe spikes, an ictal SPECT showed bi- occipital hyperperfusion, as in our patient ( 9). The relationship between the clinical and ancillary studies in our patient can be summarized as follows: 1) the transient DWI- MRI abnormalities in the right occipital lobe representing ischemia caused the right occipital seizure ( Fig. 1 A), manifested by left gaze deviation and left- beating nystagmus ( 11); 2) the seizure spread to the opposite occipital cortex, as confirmed by EEG ( Fig. IB), and as evidenced by the SPECT study showing bi- occipital hyperperfusion; 3) the bi- occipital cortical activation caused the ictal no light perception vision; and 4) the interictal surface EEG, obtained when the patient perceived light, showed high- amplitude bioccipital delta slowing and intermittent occipital, 2 Hz spike/ slow wave discharges emanating from the right occipital lobe, and accounted for the patient's lethargy and lingering light perception vision. REFERENCES 1. Barry E, Sussman NM, Bosley TM, et al. Ictal blindness and status epilepticus amauroticus. Epilepsia 1985; 26: 577- 84. 2. Visual seizures. In: Penfield W, Jasper H, eds. Epilepsy and the Functional Anatomy of the Human Brain. Boston: Little, Brown and Company, 1954; 401- 6. 3. Ludwig BI, Marsan CA. Clinical ictal patterns in epileptic patients with occipital electroencephalographic foci. Neurology 1975; 25: 463- 71. 4. Williamson PD, Thadani VM, Darcey TM, et al. Occipital lobe epilepsy: Clinical characteristics, seizure spread patterns and results of surgery. Ann Neurol 1992; 31: 3- 13. 5. Aldrich MS, Vanderzant CW, Alessi AG, et al. Ictal cortical blind-ness. Epilepsia 1989; 30: 116- 20. 6. Joseph JM, Louis S. Transient ictal cortical blindness during middle age. JNeuroophthalmol 1995; 15: 39- 42. 7. Valli G, Zago S, Capellari A, et al. Transitory and permanent visual field defects induced by occipital lobe seizures. Ital J Neurol Sci 1999; 20: 321- 5. 8. Sturm JW, Newton MR, Chinvarun Y, et al. Ictal SPECT and interictal PET in the localization of occipital lobe epilepsy. Epilepsia 2000; 41: 463- 6. 9. Bauer J, Schuler P, Feistel H, et al. Blindness as an ictal phenomenon: Investigation with EEG and SPECT in two patients suffering from epilepsy. J Neurol 1991; 238: 44- 6. 10. Jagust WJ, Budinger TF, Reed BR. The diagnosis of dementia with single photon emission computed tomography. Arch Neurol 1987: 44: 258- 62. 11. Kassem- Moussa H, Provenzale JM, Petrella JR, et al. Early diffusion- weighted MR imaging abnormalities in sustained seizure activity. AJR 2000; 174: 1304- 6. 12. Kattah JC, Potolicchio SJ, Kotz HL, et al. Cortical blindness and occipital lobe seizures induced by cisplatinum. JNeuroophthalmol-ogy 1987; 7: 99- 103. 13. Kaplan PW, Tusa RJ. Neurophysiologic and clinical correlations of epileptic nystagmus. Neurol 1993; 43: 2508- 14. 14. Kaplan PW. Neurophysiological localization of epileptic nystagmus. Am J End Technol 1999; 39: 77- 83. 26 © 2004 Lippincott Williams & Wilkins |