| OCR Text |
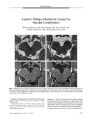
Show ORIGINAL CONTRIBUTION Radionecrosis of the Inferior Occipital Lobes With Altitudinal Visual Field Loss After Gamma Knife Radiosurgery Blythe E. Monheit, MD, John B. Fiveash, MD, and Christopher A. Girkin, MD Abstract: A patient had bilateral superior altitudinal visual field defects because of radionecrosis of the inferior occipital lobes after gamma knife radiosurgery for a recurrent atypical cerebellar meningioma. Although radionecrosis of the anterior visual pathway has been well- documented, this is the first report of visual field loss associated with occipital lobe radionecrosis. The treatment dose this patient received is within the range of predicted tolerable radiosurgical dosing, although this patient was at increased risk for radionecrosis secondary to previous external beam radiotherapy. By offering an effective, noninvasive treatment, radiosurgery has changed the management of intracranial lesions. Radiosurgery targets a discrete volume of tissue and relatively spares the surrounding normal tissue. Radiation injury, or radionecrosis, is the only significant complication of radiosurgery ( 1). We present a case of bilateral occipital lobe radionecrosis after gamma knife surgery that resulted in bilateral superior altitudinal defects. ( JNeuro- Ophthalmol 2004; 24: 195- 199) A lthough radiation necrosis of the anterior visual pathway has been amply documented, there are no reports of necrosis of the postchiasmal pathway. We report a patient who suffered bilateral superior altitudinal visual field loss after sequential external beam fractionated and single-dose radiotherapy of a cerebellar meningioma. Each treatment regimen was well within the dose limits considered nontoxic to the brain. CASE REPORT In September 2001, a 48- year- old woman was referred for neuro- ophthalmologic evaluation of sudden visual field loss present for two days and preceded by several months of intense headaches. Department of Ophthalmology ( BEM), Department of Radiation Oncology ( JBF), Department of Ophthalmology ( CAG), University of Alabama- Birmingham. Address correspondence to Blythe E. Monheit, MD, University of Alabama- Birmingham, Department of Ophthalmology, 700 18th Street S, Birmingham, AL 35233; E- mail: bmonheit@ mindspring. com An atypical cerebellar meningioma involving the straight sinus had been resected in 1992. A postoperative magnetic resonance imaging ( MRI) showed no residual tumor. However, after resection she had received external beam radiotherapy of 1.8 Gy in 33 fractions totaling 59.4 Gy. She had undergone an ophthalmologic examination in 1993 with visual acuities of 20/ 15 OU and normal threshold automated visual fields. In July 2000, a routine surveillance MRI scan demonstrated tumor recurrence ( Fig. 1), and in August 2000 the patient underwent a single gamma knife treatment of 16 Gy at the 50% isodose line. In September 2001, our examination disclosed that the patient had corrected acuities of 20/ 25 OU. All other aspects of the ophthalmologic examination were normal except that Swedish Interactive Threshold Algorithms standard 24- 2 automated visual fields showed dense superior altitudinal defects ( Fig. 2). An MRI in September 2001 showed increased enhancement of the occipital lobes bilaterally, left more than right, when compared with the scan before gamma knife treatment ( Fig. 3). A single photon emission computed tomography ( SPECT) scan illustrated reduced blood flow in the areas of occipital enhancement, consistent with radionecrosis. A review of the gamma knife treatment planning scan disclosed that the occipital lobes beneath the calcarine fissure had received radiosurgical doses near the minimum treatment dose of 16 Gy ( Fig. 4). The patient was placed on dexamethasone with no apparent improvement in the visual field defects at one- year follow- up. DISCUSSION This case is, to our knowledge, the first to report occipital lobe radionecrosis with dense superior altitudinal defects after gamma knife radiosurgery. Lars Leksell, a Swedish neurosurgeon, pioneered radiosurgery, which aimed to direct a high dose of radiation to a discrete tissue volume and to spare surrounding healthy tissue. Along withradiobiologistBorje Larsson, Leksell de- J Neuro- Ophthalmol, Vol. 24, No. 3, 2004 195 JNeuro- Ophthalmol, Vol. 24, No. 3, 2004 Monheit et al FIG. 1. Enhanced T1- weighted axial MRI scan performed in 2000, eight years after tumor extirpation and external beam radiotherapy. It shows a mass projecting from the left side of the tentorium superiorly into the supratentorial region. The patient received stereotactic radiosurgery ( 16 Gy by gamma knife at the 50% isodose line). veloped the first gamma knife unit in the 1960s ( 2). The gamma knife uses 201 cobalt sources to deliver ionizing radiation to a target volume. Its beams are collimated and convergent and may be blocked selectively to adjust the shape and volume of the tissue to be treated. The outer rim of the tumor typically receives 50% of the maximum dose (" minimal tumor dose"), which represents the 50% isodose line on the planning scan ( 1). After a stereotactic frame is placed, the patient undergoes a MRI treatment planning scan. Specialized computer software creates the plan by reconstructing the two- dimensional images into three-dimensional target volumes to be treated. The stereotactic frame serves as a reference for the coordinate system, the X-, Y-, and Z- axes, and creates the stereotactic space ( 3). There are several proposed mechanisms for radiation injury. One is damage to cellular DNA. Tumor cells have poor cell repair mechanisms and are, relative to normal cells, unable to recover from the radiation. Normal cells may be damaged if the dose they receive is too high ( 4). Tandon et al ( 5) believe that injury is based in the vascular endothelium; radiation increases the production of coagulative factors that cause vascular thrombosis. At the same time, the cell cycle of the endothelium is disrupted, which causes a large number of abnormally functioning endothelial cells to be produced, with subsequent breakdown in the blood- brain barrier. Other studies have examined the role of cytokines in radiation injury. Kureshi et al ( 6) found in-terleukin- 1, tumor necrosis factor alpha, and interleukin- 6 expressed by infiltrating macrophages during radionecrosis of cerebral tissue. Damage to healthy tissue surrounding the treated lesion may appear months to years after focal brain irradiation ( 2). The presenting symptom is usually a headache, focal or generalized seizures, or hemiparesis. MRI reveals a hypointense mass, sometimes with contrast enhancement, which is not readily distinguishable from malignant tissue. Biopsy is diagnostic, demonstrating coagulative necrosis of white matter, telangiectasia, fibrinoid necrosis of blood vessels, thrombus formation, glial cell proliferation, and multinucleated giant astrocytes. When biopsy is not possible, SPECT or positron emission tomography scan with radio- labeled glucose will show decreased metabolism in the area of interest, which suggests radionecrosis rather than recurrent malignancy ( 7). Visual field defects have been well described after the use of conventional external beam therapy to lesions near the anterior visual pathway, including pituitary tumors, craniopharyngiomas, parasellar meningiomas, and optic nerve meningiomas ( 8- 12). The defects generally occur one or more years after radiation treatment. On MRI, there is optic nerve swelling and enhancement ( 13). Radiation optic neuropathy after gamma knife therapy has been less * FIG. 2. Humphrey visual fields performed in 2001, one year after the patient had received stereotactic radiosurgery to the recurrent cerebellar tumor. They show superior alti-tudinal defects reflecting damage to inferior visual cortical regions. 196 © 2004 Lippincott Williams & Wilkins Radionecrosis of the Inferior Occipital Lobes JNeuro- Ophthalmol, Vol. 24, No. 3, 2004 FIG. 3. MRI performed at the time of detection of superior altitudinal visual field loss. Enhanced axial T1- weighted scans ( A, B, C) demonstrate enhancement of the white matter of the occipital lobes, left more than right. Axial T2- weighted scans ( D, E, F) show high signal in the occipital white matter. Reduced blood flow on the SPECT scan in this region confirmed the impression of radionecrosis. often reported perhaps because patients with lesions near the optic structures are less often treated with single fraction radiotherapy. Yet, a 1997 series reported 2400 patients treated by gamma knife for lesions around the anterior visual pathway, ( 2) and only four ( 0.2%) had visual loss up to 30 months later. Delivery of more than 8 Gy was associated with a higher incidence of tissue injury. Additional risk factors for developing radiation damage included previous external beam radiotherapy and visual dysfunction before treatment ( 2). In a series of patients treated for cavernous sinus lesions with single fraction stereotactic radiation, Leber et al ( 23) found the incidence of optic neuropathy to be zero in patients exposed to 10 Gy or less. Doses greater than 10 Gy resulted in dramatic increases in optic neuropathies, with 15 Gy having a 77.8% incidence. In contrast, the oculomotor and trigeminal nerves were much more tolerant to the same doses ( 23). In another series of 66 patients who had cavernous sinus and petroclival area lesions treated by gamma knife ( 14), the optic nerves, chiasm, or tract received between 1 and 16 Gy, with a median of 10 Gy. In 59 ( 89%) of cases, the dose was greater than 8 Gy, yet there was no visual loss after a two- year follow up. The authors postulated that the anterior visual pathway may tolerate a dose of 12 to 16 Gy provided there are no factors that would predispose the patient to radionecrosis, including previous dysfunction or previous radiosurgery at the same site ( 14). Guidelines to estimate the risk of cortical radionecrosis after gamma knife treatment have been actively pursued. The foundation of radiosurgical prescription dosing today is based on work performed by Raymond Kjellberg in 1983 ( 15). His study centered on the dose of Gy delivered to a tissue volume as the predictor of radionecrosis. He formulated a curve of volume of tissue treated versus dose of Gy that would predict a 1% chance of radionecrosis ( 15). Chin et al ( 1) suggest a dose of 10 Gy given to a volume of tissue 10 mm3 or more is associated with a higher risk of radionecrosis ( 1). Flickinger et al ( 16,18) found that the risk of radionecrosis after gamma knife radiosurgery in patients with arteriovenous malformations varied dramatically with location within the brain and volume of tissue receiving 12 Gy. Other studies suggest that " conformity," the ability of the treatment to spare surrounding normal tissue, is an important predictor in radiation injury. The ratio of prescription volume ( total treated volume) to target volume ( treated tumor volume) is the conformity index. In radiosurgery, this index should be less than two ( 17). The studies on the risk of radionecrosis from radiosurgery have reached the following conclusions: 1) the most important predictors of radionecrosis are tumor vol- 197 JNeuro- Ophthalmol, Vol. 24, No. 3, 2004 Monheit et al FIG. 4. The gamma knife planning scan of the patient. The three- dimensional reconstruction of the target tumor and surrounding tissue volumes shows isodose lines around the margins of the tumor, with the 50% isodose line representing the enhancing tumor margin, which will receive 50% of the maximum tumor dose. ume, treated volume, total dose, and conformity ( see below); and 2) a subset of " vulnerable" patients develop radi-onecrosis at much lower levels than normally observed ( 1,18). Ongoing research is aimed at identifying these vulnerable patients to appropriately modify treatment ( 19). Our patient had external beam radiotherapy ( EBRT) before her gamma knife treatment, which increased her risk for radionecrosis. This variable is not included in the aforementioned studies. Stafford et al ( 24) evaluated the tolerance of the optic apparatus to gamma knife after stereotactic radiosurgery. Among 215 patients with benign tumors adjacent to the anterior optic apparatus treated with gamma knife radiosurgery, 23 had had previous EBRT with a mean dose of 50.2 Gy. One patient had EBRT in conjunction with radiosurgery. Three of the four patients who had radionecrosis after gamma knife radiosurgery had previous EBRT or concurrent EBRT. The authors concluded that the risk of radionecrosis in patients receiving a point dose of 12 Gy or less is 1%, but that the risk increases to 13% if the patient has had previous EBRT ( 24). Shaw et al ( 25) studied maximum tolerated doses of single fraction radiosurgery in patients with recurrent, previously irradiated primary brain tumors and metastases. In that study, 156 patients had received a median dose of 60 Gy from previous fractionated radiation therapy and were later treated with stereotactic radiosurgery. The patients were divided by tumor size: 1) small ( less than 20 mm diameter with a median volume of 3.6 mT); 2) medium ( 21- 30 mm diameter with median volume of 6 mT); and 3) large ( 31- 40 mm diameter with median volume of 17.9 mT). The authors found that the maximum tolerable doses of single fraction radiosurgery in patients with previously irradiated tumors to be 24 Gy in small tumors, 18 Gy in medium- sized tumors, and 15 Gy in large tumors. These doses were prescribed to the 50% to 90% isoline, which encompassed the entire enhancing tumor volume. Patients with medium- sized tumors ( 21- 30 mm) had 7.3- fold higher risk and those with large tumors ( 31- 40 mm) had a 16- fold higher risk for unacceptable central nervous system toxicity than patients with small tumors (< 20 mm) ( 25). In our patient, the treatment plan ( Fig. 4) covered the enhancing margins of the meningioma with 16 Gy at the 50% isoline. The maximum dose was 32 Gy, delivered to the center of the lesion, and the 25% isoline, which encompassed the occipital lobes, was 8 Gy. The tumor volume treated was 7.9 cm3; the perilesional tissue treated was 7.6 cm3. Thus, the total volume of tissue treated was 15.5 cm3. This volume received greater than or equal to 16 Gy. With 198 © 2004 Lippincott Williams & Wilkins Radionecrosis of the Inferior Occipital Lobes JNeuro- Ophthalmol, Vol. 24, No. 3, 2004 a tumor volume of 7.9 mL, our patient would be included in the medium- sized tumor group of Shaw et al ( 25). By their calculations, the maximum tolerable dose would be 18 Gy to the 50% isoline, greater than the estimated 16 Gy dose she received. Thus, she received a " tolerable" dose as predicted by the aforementioned study. She did have, however, a 7.3- fold higher risk for radionecrosis than a patient with a smaller tumor. At this time, there is no proven treatment of radionecrosis. There are several classes of drugs, all of which have been used with variable response. Corticosteroids have been used to decrease cerebral edema ( 1) and the production of cytokine release after radiation, but have not proven to change the clinical course of radionecrosis. Lazaroids ( 21- aminosteriods) are effective in animal models ( 5), but human data do not exist. Hyperbaric oxygen therapy is thought to be helpful by raising the tissue p02 and initiating a cellular and vascular repair. One study based in Austria by Leber et al ( 20) evaluated the effects of hyperbaric oxygen therapy on two patients with signs of radionecrosis after gamma knife treatment. The two patients were treated by breathing 100% oxygen at 2.5 atm absolute for one hour each day, 40 times in cycles of 10 sessions. In this study, one patient had resolution of the radiographic findings of tissue injury, whereas the other had a reduction in the size of the damaged area. No corticosteroids were given to these patients ( 20). The efficacy of hyperbaric oxygen has been questioned by Brown et al ( 21) and Roden et al ( 22). In summary, we present a case of radionecrosis of the occipital cortex resulting in visual field loss after gamma knife radiosurgery to a recurrent cerebellar tumor. We have reviewed the radiosurgical literature and applied predicative models to our patient to retrospectively assess her risk of this complication. The radiosurgical dose she received was within the predicted tolerable range but she carried a 7.3 fold increased risk of radionecrosis with this dose than that of a patient with a smaller tumor. REFERENCES 1. Chin LS, Ma L, DiBiase S. Radiation necrosis following gamma knife surgery: a case- controlled comparison of treatment parameters and long- term clinical follow up. JNeurosurg 2001; 94: 899- 904. 2. Girkin CA, Comey CH Lunsford LD. Radiation Optic Neuropathy after Stereotactic Radiosurgery. Ophthalmology 1997; 104: 1634^ 13. 3. Weil MD. Stereotactic Radiosurgery for Brain Tumors 2001 ; 15: 1017- 26. 4. Rakel RE, Bope ET. Conn's Current Therapy, 54th ed. Philadelphia: WB Saunders; 2002. 5. Tandon N, Vollmer DG, New PZ. Fulminant radiation- induced necrosis after stereotactic radiation therapy to the posterior fossa. Case report and review of the literature. J Neurosurg 2001; 95: 507- 12. 6. Kureshi SA, Hofman FM, Schneider JH. Cytokine Expression in Radiation- induced Delayed Cerebral Injury. Neurosurgery 1994: 35: 822- 30. 7. Goldman L, Bennett JC. Cecil Textbook ofMedicine, 21st ed. Philadelphia: WB Saunders; 2000. 8. Buys NS, Kerns TC Jr. Irradiation Damage to the Chiasm. Am J Ophthalmol 1957; 44: 483- 6. 9. Schatz NJ, Lichtenstein S, Corbett JJ. Delayed radiation necrosis of the optic nerves and chiasm. In: Glaser JS, Smith JL, eds. Neuro-ophthalmology Symposium of the University of Miami and the Bas-com Palmer Eye Institute, Vol 8. St. Louis: CVMosby; 1978: 131- 9. 10. Hammer HM. Optic chiasmal radionecrosis. Trans OphthalmolSoc UK 1983; 103: 208- 11. 11. Kline LB, Kim JY, Ceballos R Radiation Optic Neuropathy. Ophthalmology 1985; 92: 1118- 26. 12. Warman R, Glaser JS, Quencer RM et al. Radionecrosis of optico-hypofhalamic glioma. Neuroophthalmology 1989; 9: 219- 26. 13. Guy J, Mancuso A, Quisling RG, et al. Gadolinium- DTPA- enhanced magnetic resonance imaging in optic neuropathies. Oph-thamology 1990; 97: 592- 600. 14. Morita A, Coffey RJ, Foote RL. Risk of Injury to cranial nerves after gamma knife radiosurgery for skull base meningiomas; experience in 88 patients. J Neurosurg 1999; 90: 42- 9. 15. Kjellberg RN, Hanamura T, Davis KR et al. Bragg- peak proton-beam therapy for arteriovenous malformations of the brain. N Engl J Med 1983; 309: 269- 74. 16. Flickinger JC. An integrated logistic formula forprediction of complications from radiosurgery. Int J Rod One Biol Phys 1989; 17: 879- 85. 17. Voges J, TreuerH, SturmVetal. Risk analysis of linear accelerator radiosurgery. Int JRad One Biol Phys 1996; 36: 1055- 63. 18. Flickinger JC, Kondziolka D, Lunsford LD et al. Development of a model to predict permanent symptomatic postradiosurgery injury for arteriovenous malformation patients. Int J Rad One Biol Phys 2000; 46: 1143- 8. 19. Nakamura JL, Verhey LJ, Smith V et al. Dose Conformity of Gamma Knife Radiosurgery and Risk Factors For Complications. Int J Rad One Biol Phys 2001; 51: 1313- 9. 20. Leber KA, Eder HG, Kovac H. Treatment of Cerebral Radionecrosis by hyperbaric oxygen therapy. Stereotac FunctNeurosurg 1998: 70: 229- 36. 21. Brown GC, Shields JA, Sanborn G, et al. Radiation Optic Neuropathy. Ophthalmology 1982; 89: 1489- 93. 22. Roden D, Bowsley TM, Fowble B, et al. Delayed Radiation injury to the retrobulbar optic nerves and chiasm: Clinical syndrome and treatment with hyperbaric oxygen and corticosteroids. Ophthalmology 1990; 97': 346- 51. 23. Leber KA, Bergloff J, Pendl G. Dose Response Tolerance of the Visual Pathways and Cranial Nerves of the Cavernous Sinus to Stereotactic Radiosurgery. J Neurosurgery 1998; 88: 43- 50. 24. Stafford SL, Pollock BE, Leavitt JA, et al. A Study on the Radiation Tolerance of the Optic Nerves and Chiasm After Stereotactic Radiosurgery. Int. J. Radiation Oncology Biol Phys 2003; 55: 1177- 81. 25. Shaw E, Scott C, Souhami L. Single Dose Radiosurgical Treatment of Recurrent Previously Irradiated Primary Brain Tumors and Brain Metastases: Final Report of RTOG Protocol 90- 05. Int J Radiat Oncology Biol Phys 2000; 47: 291- 8 199 |