| Description |
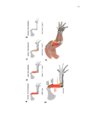
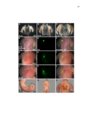
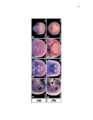
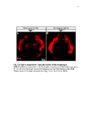
Development of the limb musculoskeleton requires the coordinated morphogenesis of muscle with connective tissue, tendon, and bone. The close developmental association of muscle with muscle connective tissue suggests that interactions between these tissues may be critical for development. T-box transcription factors have been shown to non-cell autonomously regulate limb muscle development. Mutations in TBX3 lead to ulnar-mammary syndrome, a congenital disorder with posterior limb defects. However, identification of the role of Tbx3 in limb development has previously been hindered by early embryonic lethality. Using mouse genetics and conditional mutagenesis, we identify the Tbx3 cell lineage and determine its role in musculoskeletal development. We show that Tbx3 gives rise to non-muscle cells in the developing forelimb. We identify a cell autonomous role for Tbx3 in ulna and bone eminence development. We also identify a non-cell autonomous role for Tbx3 in muscle fiber formation. Finally, our studies in mouse have led us to uncover previously undiagnosed skeletal and muscle defects associated with ulnar-mammary syndrome. Thus, Tbx3 plays multiple, critical roles in forelimb development. The muscularized diaphragm is a defining characteristic of all mammals. However, the developmental innovation(s) leading to the evolution of this structure remain unknown. Muscularization of the diaphragm requires migration of muscle cells into the pleuroperitoneal folds. Congenital diaphragmatic hernia, a lethal birth defect, suggests that partial muscularization would be deleterious and that evolution of the muscularized diaphragm likely resulted from a simple genetic modification that rapidly fixed in the population. Through comparison of mammalian diaphragm development with non-mammalian septum development, I have identified developmental differences that suggest Hgf expression in the pleuroperitoneal folds may underlie the evolution of the mammalian diaphragm. I have determined that PPF-like structures are present in the chick; however, they are void of muscle cells and do not express Hgf. I have also shown that Hgf expression in the pleuroperitoneal folds is critical for complete muscularization of the diaphragm. Thus, I have determined that Hgf is required for development of the diaphragm and that pleuroperitoneal fold-specific expression of Hgf may have contributed to the evolution of the mammalian diaphragm. |