| OCR Text |
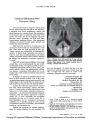
Show ORIGINAL CONTRIBUTION Bilateral Optic Neuritis in Acute Hepatitis C Junaid Siddiqui, MD, Jacinthe Rouleau, MD, Andrew G. Lee, MD Yutaka Sato, MD, and Michael D. Voigt, MD Abstract: A 34-year-old woman developed bilateral optic neuritis 2 weeks after the onset of acute hepatitis C. The strong temporal relationship between the initial clinical manifestations of hepatitis C and the development of optic neuritis provides a basis for thinking that the hepatitis caused the optic neuritis After corticosteroid treatment, the optic neuropathy markedly improved but left behind retinal nerve fiber thinning, as measured by optical coherence tomog-raphy, and optic disc pallor. Optic neuritis has been reported in conjunction with hepatitis A and B but not with hepatitis C. (J Neuro-Ophthalmol 2009;29:128-133) Postinfectious optic neuritis occurs in association with a number of bacterial and viral infections, including those due to adenovirus, Coxsackie virus, cytomegalovirus, Epstein-Barr virus, human immunodeficiency virus, mea-sles, mumps, rubeola, rubella, varicella and herpes zoster, and rarely hepatitis A (1,2) and B (3-5). Optic neuritis has not been reported in association with acute hepatitis C, a major global health problem that affects an estimated 170 million people worldwide. It causes chronic hepatitis, which may lead to cirrhosis in up to 20% of chronically infected individuals, and is now the leading indication for liver transplantation (6). Frequently asymptomatic, it can also present with acute severe hepatitis and extrahepatic complications (7,8). We report a case of bilateral optic neuritis in a patient with acute hepatitis C. CASE REPORT A 34-year-old woman presented with jaundice, dark urine, myalgia, nausea, vomiting, and dysuria. Serum Departments of Gastroenterology and Hepatology (JS, MDV), Neuro-Ophthalmology (JR), Radiology (AGL), and Ophthalmology, Neurology, and Neurosurgery, The H. Stanley Thompson Neuro- Ophthalmology Clinic (YS), University of Iowa Hospitals and Clinics, Iowa City, Iowa. Address correspondence to Michael D Voigt, MD, 4553 JCP, 200 Hawkins Dr., Iowa City, IA 52242; E-mail: michael-voigt@uiowa.edu transaminase levels were markedly elevated in keeping with acute hepatitis (Fig. 1). She had not used any hepatotoxic medications or acetaminophen. Hepatitis C virus (HCV) antibodies were positive, and HCV RNA was present at a level of 300 IU/ml with genotype la. Results for hepatitis B surface antigen, hepatitis B core IgM antibodies, autoimmune hepatitis markers (antinuclear, anti-mitochondrial, anti-smooth mus-cle, and anti-atypical P-neutrophil cytoplasmic antibodies), cytomegalovirus DNA, and acute antibodies to Epstein- Barr virus were all negative. Liver ultrasound showed a normal-sized liver and spleen. Liver biopsy (Fig. 2) showed a heavy infiltrate of lymphocytes in the portal and lobular areas in keeping with acute hepatitis. Klatskin stain showed normal architecture and minimal fibrosis. A diagnosis of acute hepatitis C was established. Fourteen days after the onset of these symptoms, she developed a headache, her eyes became red and she reported worsening vision in both eyes. Visual acuity was 20/200 in the right eye and 20/50 in the left eye with pinhole at a bedside examination. Pupils measured 4 mm in the dark and 3 mm in the light with a 0.3-log unit relative afferent pupil defect in the right eye. Ocular motility and alignment were normal. The anterior ocular segment appeared normal. There was mild optic disc edema in both eyes. Goldmann perimetry showed dense cecocentral scotomas in both eyes (Fig. 3A). Results of neurologic examination were otherwise normal. Retinal fluorescein angiography showed bilateral optic disc hyperfluorescence consistent with optic disc edema. Optical coherence tomography (OCT) of the peripapillary retina using the fast retinal nerve fiber layer thickness protocol showed an average thickness of 138 um in the right eye and 107 um in the left eye (normal 100 ± 11 um). Brain MRI showed T2 high signal, enlargement, and enhancement of the intraorbital and intracranial segments of both optic nerves and enhancement of the optic chiasm without other abnormalities (Fig. 4). Results of chest x-ray, complete blood count (CBC), erythrocyte sedimentation rate (ESR), and serum protein electrophoresis; serum rapid plasma reagin (RPR), Lyme disease titer, toxoplasma, and Bartonella henselae anti-bodies; viral studies for cytomegalovirus, Epstein-Barr 128 J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. FIG. 1. Time course of biochemical changes of the acute hepatitis C. Day 0 was the day of the first medical encounter. Visual symptoms antedated day 0 by 3 weeks. virus, enterovirus and herpes simplex, and serum double- immunofixation electrophoresis revealed a CSF IgG of stranded DNA, cryoglobulins, and rheumatoid factor were 8% (normal 0-14%), a CSF IgG index of 0.5 (normal 0.0- normal or negative. Serum angiotensin-converting enzyme 0.7), and de novo synthesis of 1.7mg/day (normal 9.9 to (ACE) was marginally elevated at 58 U/L (normal 3-52 3.3 mg/day). U/L), but she had no other clinical or chest X-ray findings She was treated with 1 g intravenous methylpred-compatible with sarcoidosis. Cerebrospinal fluid (CSF) nisolone daily for 3 days followed by a 14-day oral taper, analysis was normal, including VDRL and fluorescent Six weeks after the corticosteroid treatment was started and treponemal antibodies. Paired serum and CSF before the start of HCV treatment, visual acuity had FIG. 2. Liver biopsy demonstrates dense portal lymphocytic infiltrate (black arrows), interface hepatitis (white arrow), preserved architecture (right inset), and liposome-laden sinusoidal macrophages (black arrowheads, left inset). (Hematoxylin and eosin, X400; right inset Klatskin stain, X100; left inset periodic acid Schiff stain, X400.) 129 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. Optic Neuritis in Hepatitis C J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 Siddiqui et al FIG. 3. A. Goldmann perimetry at presentation shows dense cecocentral scotomas with breakout inferiorly in both eyes. B. Goldmann perimetry performed 14 weeks later shows improvement after treatment with corticosteroids. improved to 20/40 in the right eye and 20/30 in the left eye. acute hepatitis had resolved. She had no detectable serum Pegylated interferon and ribavirin therapy for hepatitis C HCV RNA after completion of pegylated interferon and was started 72 days after the onset of her initial symptoms ribavirin therapy, of acute hepatitis and approximately 8 weeks after the initial ophthalmologic examination (Fig. 1). Follow-up examination at 4 months after presentation DISCUSSION showed that her vision had markedly improved to 20/20 in The striking feature of this case is the clear temporal both eyes, but both optic discs were pale (Fig. 5). relationship between the onset of acute hepatitis C and optic Goldmann visual fields were improved (Fig. 3B). Repeat neuritis, making a strong case for hepatitis C as the cause. OCT confirmed thinning of the retinal nerve fiber layer with There have been prior reports of optic neuritis measurements of 64 um in the right eye and 60 um in the associated with acute hepatitis A and B, but not hepatitis C left eye. Seven months after the initial presentation, her (Table 1). Jouhadietal(l) described a case of neuromyelitis FIG. 4. Brain MRI at presentation. Precontrast (A) and postcontrast (B) coronal T1 studies show enhancement of both intraor-bital optic nerves (arrows). Coronal T2 study (C) shows an abnormally high signal in both intraorbital optic nerves (arrows). 130 © 2009 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. Optic Neuritis in Hepatitis C J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 131 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. with bilateral simultaneous papillitis in an 8-year-old child after acute viral hepatitis A infection. An additional case of bilateral optic neuritis after hepatitis A with good visual recovery was reported (2). Achiron (3) presented a case of bilateral optic neuritis associated with systemic vasculitis and glomerulonephritis after acute hepatitis B. Galli et al (5) described a unilateral retrobulbar optic neuritis in a 33- year-old woman with acute hepatitis B. Albitar et al (9) reported a case of retrobulbar optic neuritis after recom-binant hepatitis B vaccination in a patient with end-stage renal disease. In prior cases of viral hepatitis-associated optic neuritis, four of five cases were bilateral with optic disc edema at onset, whereas one case of hepatitis B was associated with unilateral retrobulbar optic neuritis. The time from the onset of the systemic viral symptoms to the ophthalmic symptoms in our patient was similar to that of previously reported patients (7-40 days) and she seemed to have a similar favorable response to high-dose corticosteroid treatment. However, although visual symptoms improved or resolved in all cases after corticosteroid treatment, all patients, with the exception of the patient of Jouhadi et al (1), were left with impairment of visual acuity, color vision, or visual field or pallor of the optic discs. MRI abnormalities in our patient were similar to those reported by McKibbin in hepatitis A (2). However, MRI was reported to be normal in three other reports (Table 1) (1,3,9), two of which appeared in the mid-1990s when imaging technology was less sensitive. The mechanism for optic neuritis in patients with acute hepatitis is unknown. Complement activation and autoimmune-mediated neurotoxicity have been proposed. Galli et al (5) described a unilateral retrobulbar optic neuritis in a 33-year-old woman with acute hepatitis B and found that complement activation, involving both classic and alternative pathways, and high levels of circulating immune complexes were present at the onset of ophthalmic symptoms. HCV is associated with autoimmune or lymphopro-liferative states perhaps related to the propensity of the virus to replicate in lymphoid cells (10). Mixed cryoglobulinemia associated with hepatitis C can result in vasculitis, causing weakness, arthralgia, purpura, glomeru-lonephritis, and mononeuritis multiplex (11,12). Cryoglo-bulinemia may cause cerebral vasculitis (13-19) or leukoencephalopathy (20). However, there was no evidence clinically, by fluorescein angiography, or by brain imaging of vasculitis in our patient, and she did not have cryoglobu-linemia. There is evidence that central nervous system (CNS) replication of HCV occurs (21-23), possibly because mutated viruses have developed adaptations that allow them to proliferate in neural tissue (24). Hepatitis C is associated with altered cytokine profiles in CSF (20). Neurotropism and an altered immune response could possibly account for inflammation, leading to immune-mediated demyelination (25). Deposit of immune complexes on the myelin sheath or the cross-reaction of antibodies against myelin basic protein could impair myelin function (11). The minor elevation in serum ACE level was thought to be non-specific (26), as there were no clinical or radiological features to support the diagnosis of sarcoidosis. We reject multiple sclerosis as the explanation for the optic neuritis in our patient because the optic neuritis was simultaneously bilateral and unassociated with demyelin-ating white matter lesions on MRI or CSF changes compatible with multiple sclerosis. Treatment of hepatitis C may cause bilateral anterior ischemic neuropathy (27,28), retinal artery or vein occlusion (29-32), macular edema, or nonmacular retinopathy at any time in the course in inter-feron therapy (33-37). The most frequent abnormalities are cotton wool spots and retinal hemorrhages (36,38,39) with a higher incidence in patients with concurrent hypertension (40) or diabetes (41). We do not think that treatment of hepatitis C played a role in our patient because it was started after her optic neuropathy had improved and there was continued recovery while she was receiving treatment. Acknowledgment We thank Dr. Jamie Weydert for interpretation of the liver pathologic samples. REFERENCES 1. Jouhadi Z, Ouazzani I, Abid A, et al. Devic's optic neuromyelitis and viral hepatitis type A: a paediatric case report (in French). Rev Neurol (Paris) 2004;160:1198-202. 2. McKibbin M, Cleland PG, Morgan SJ. Bilateral optic neuritis after hepatitis A. J Neurol Neurosurg Psychiatry 1995;58:508. 3. Achiron LR. Postinfectious hepatitis B optic neuritis. Optom Vis Sci 1994;71:53-6. 4. Farthing CF, Howard RS, Thin RN. Papillitis and hepatitis. Br Med J (Clin Res Ed) 1986;292;1712. 5. Galli M, Morelli R, Casellato A, et al. Retrobulbar optic neuritis in a patient with acute type B hepatitis. J Neurol Sci 1986;72:195-200. 6. Patel K, Muir AJ, McHutchison JG Diagnosis and treatment of chronic hepatitis C infection. BMJ 2006;332:1013-7. J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 Siddiqui et al 132 © 2009 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. FIG. 5. Fundus photographs show mild optic disc pallor temporally in each eye. Optic Neuritis in Hepatitis C J Neuro-Ophthalmol, Vol. 29, No. 2, 2009 7. Cox AL, Netski DM, Mosbruger T, et al. Prospective evaluation of comparative analysis of internal translational efficiency of brain, community-acquired acute-phase hepatitis C virus infection. Clin liver, and serum variants. J Virol 2004;78:5170-83. Infect Dis 2005;40:951-8. 25. Weiner HL, Hauser SL. Neuroimmunology. II: Antigenic specificity 8. Wang CC, Krantz E, Klarquist J, et al. Acute hepatitis C in of the nervous system. Ann Neurol 1982;12:499-50. a contemporary US cohort: modes of acquisition and factors 26. Muller BR. Analysis of serum angiotensin-converting enzyme. Ann influencing viral clearance. J Infect Dis 2007;196:1474-82. Clin Biochem 2002;39:436-43. 9. Albitar S, Bourgeon B, Genin R, et al. Bilateral retrobulbar optic 27. Willson RA. Visual side effects of pegylated interferon during neuritis with hepatitis B vaccination. Nephrol Dial Transplant 1997; therapy for chronic hepatitis C infection. J Clin Gastroenterol 2004; 12:2169-70. 38:717-22. 10. Lauer GM, Walker BD. Hepatitis C virus infection. N Engl J Med 28. Gupta R, Singh S, Tang R, et al. Anterior ischemic optic 2001;345:41-52. neuropathy caused by interferon alpha therapy. Am J Med 2002; 11. Heckmann JG, Kayser C, Heuss D, et al. Neurological manifestations 112:683-4. of chronic hepatitis C. J Neurol 1999;246:486-91. 29. Nicolo M, Artioli S, La Martina GC, et al. Branch retinal artery 12. Kayali Z, Buckwold VE, Zimmerman B, et al. Hepatitis C, occlusion combined with branch retinal vein occlusion in a patient cryoglobulinemia, and cirrhosis: a meta-analysis. Hepatology with hepatitis C treated with interferon and ribavirin. Eur J 2002;36:978-85. Ophthalmol 2005;15:811-4 13. Arena MG, Ferlazzo E, Bonanno D, et al. Cerebral vasculitis in 30. Akyuz F, Akyuz U, Kocaman O, et al. Rare complication of interferon a patient with HCV-related type II mixed cryoglobulinemia. J Investig therapy: retinal vein thrombosis. Acta Gastroenterol Belg 2005 ;68: Allergol Clin Immunol 2003;13:135-6. 394-5. 14. Cappellari A, Origgi L, Spina MF, et al. Central nervous system 31. Kiratli H, Irkec M. Presumed interferon-associated bilateral macular involvement in HCV-related mixed cryoglobulinemia. Electromyogr arterial branch obstruction. Eye 2000;14:920-2. Clin Neurophysiol 2006;46:149-58. 32. Nadir A, Amin A, Chalisa N, et al. Retinal vein thrombosis 15. Casato M, Saadoun D, Marchetti A, et al. Central nervous system associated with chronic hepatitis C: a case series and review of the involvement in hepatitis C virus cryoglobulinemia vasculitis: literature. J Viral Hepat 2000;7:466-70. a multicenter case-control study using magnetic resonance imaging 33. Ogata H, Suzuki H, Shimizu K, et al. Pegylated interferon-associated and neuropsychological tests. J Rheumatol 2005;32:484-8. retinopathy in chronic hepatitis C patients. Jpn J Ophthalmol 2006; 16. Dawson TM, Starkebaum G. Isolated central nervous system vasculitis 50:293-5. associated with hepatitis C infection. J Rheumatol 1999;26:2273-6. 34. Tu KL, Bowyer J, Schofield K, et al. Severe interferon associated 17. Fragoso M, Carneado J, Tuduri I, et al. Essential mixed retinopathy. Br J Ophthalmol 2003;87:247-8. cryoglobulinemia as a cause of ischemic cerebrovascular accident 35. Neubauer AS, Hoops JP Interferon-induced retinopathy in asymp- (in Spanish). Rev Neurol 2000;30(5): 444-6. tomatic cancer patients. Ophthalmology 2002;109:821-2. 18. Origgi L, Vanoli M, Carbone A, et al. Central nervous system 36. Jain K, Lam WC, Waheeb S, et al. Retinopathy in chronic hepatitis C involvement in patients with HCV-related cryoglobulinemia. Am J patients during interferon treatment with ribavirin. Br J Ophthalmol Med Sci 1998;315:208-10. 2001;85:1171-3. 19. Propst T, Propst A, Nachbauer K, et al. Papillitis and vasculitis of the 37. Suzuki T, Yonemura K, Miyaji T, et al. Progressive renal failure and arteria spinalis anterior as complications of hepatitis C reinfection blindness due to retinal hemorrhage after interferon therapy for after liver transplantation. Transpl Int 1997;10:234-7. hepatitis C virus-associated membranoproliferative glomerulone- 20. Buccoliero R, Gambelli S, Sicurelli F, et al. Leukoencephalopathy as phritis. Intern Med 2001;40:708-12. a rare complication of hepatitis C infection. Neurol Sci 2006;27:360-3. 38. Hayasaka S, Fujii M, Yamamoto Y, et al. Retinopathy and 21. Laskus T, Radkowski M, Adair DM, et al. Emerging evidence of subconjunctival haemorrhage in patients with chronic viral hepatitis hepatitis C virus neuroinvasion. AIDS 2005;19:S140-4. receiving interferon alfa. Br J Ophthalmol 1995;79:150-2. 22. Morsica G, Bernardi MT, Novati R, et al. Detection of hepatitis C 39. Guyer DR, Tiedeman J, Yannuzzi LA, et al. Interferon-associated virus genomic sequences in the cerebrospinal fluid of HIV-infected retinopathy. Arch Ophthalmol 1993;111:350-6. patients. J Med Virol 1997;53:252-4 40. Okuse C, Yotsuyanagi H, Nagase Y, et al. Risk factors for retinopathy 23. Radkowski M, Wilkinson J, Nowicki M, et al. Search for hepatitis C associated with interferon alpha-2b and ribavirin combination virus negative-strand RNA sequences and analysis of viral sequences therapy in patients with chronic hepatitis C. World J Gastroenterol in the central nervous system: evidence of replication. J Virol 2002; 2006;12:3756-9. 76:600-8. 41. Willson RL, Ross RD, Willson LM, et al. Interferon-associated 24. Forton DM, Karayiannis P, Mahmud N, et al. Identification of unique retinopathy in a young, insulin-dependent diabetic patient. Retina hepatitis C virus quasispecies in the central nervous system and 2000;20:413-5. 133 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. |