| OCR Text |
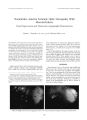
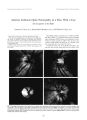
Show :-<> l'lllll, llinol,> « V VVilkms, P h i l a d e l p h ia Transient Cortical Blindness in Preeclampsia With Indication ol Generalized Vascular Endothelial Damage Anat Kesler. M. D.. Ha » ai Kancti. M. D.. and Dcvora Kidron. M. D. A 26- ycar- old woman with twin Ictuses of 2< X weeks' gcsla-tional age had symploms of preeclampsia and was admitted to the hospital for ohserv ation. Nine days later, after reporting a severe hcailaehe. the patient experienced loss of vision in both eyes. An emergency computed tomographic brain scan was performed to rule out intracranial hemorrhage, and cesarean delivers was performed. Twelve hours after the operation, the patient's vision improved gradually and returned to normal after 24 hours. The placenta was submitted to pathologic examination, and" magnetic resonance imaging was performed - I ilavs after birth. Recent thrombosis observed in the histologic section of the placenta, ischemic changes in the brain seen in the computed tomographic and magnetic resonance scans, and severe proteinuria manifested clinically suggest vascular endothelial damage as the undcrlviug mechanism in this case of preeclampsia- relaled transient cortical blindness. Key Words: Cortical blindness- Preeclampsia laidolhelial damasic. Preeclampsia is a hypertensive disorder of late pregnancy that resolves shortly alter delivery. It occurs in as much ; is I0' 7r of till pregnancies, most often in primi-paras, and is a major cause of maternal and fetal morbidity ( I - 3). Recognized as a specific disorder since the time of Hippocrates, the only completely successful therapy remains the delivery of the placenta ( 4). Ophthalmic manifestations of preeclampsia include retinal arteriolar spasm, optic neuropathy, serous retinal detachment, and cerebral blindness, more commonly termed cortical blindness ( 2,5,6). Isolated cortical blindness associated with preeclampsia is unusual. It had been thought to occur in only I '-< to 3' A of pregnancies ( 7 9), but has recently been reported to occur in as many as 15'/ r of cases ( 10). The present report concerns a ease o( blindness in a woman with preeclampsia, in which the presence of occipital lobe cerebral edema and typical changes are suggested by computed tomography ( C'l'l and magnetic resonance imaeini: ( MRI). A detailed Manuscript received August I')')'/; accepted April l')(), S. I; mm the Departments of Neurologv ( A. K. i. Obstetrics ill. K. i. and ( ivnecologv. IVrinalal I int. Pathology ID. K. I, Men Medical (' enter. Kl. ir Saba. Israel. Address correspondence and reprint requests to Dr. Anat Keslci. Department of Neurology. Men Medical Center. Kfar Saba 442KI. pathologic examination of the placenta was performed in tin attempt to contribute to the understanding of the underlying mechanism of this rare entity. CASK RKPORT A 26- year- old woman ( gravida 3. parti I), with twin fetuses of 2S weeks' gestational age had symptoms of preeclampsia: blood pressure. 130/ 90; urine dipstick. 4- 4 protein; and mild peripheral edema. Gynecologic history w; ts of primary and secondary amenorrhea, probably caused h\ irregular ovulation. The first pregnanes' occulted after treatment with clomiphene citrate ( lkaclo-inin, Teva Pharmaceuticals, Israel) to induce ovulation and resulted in a 3035- g full- term female infant: the second pregnancy ended in miscarriage in the first trimester, followed by dilation and curettage; and the third anil present pregnancy, also induced by clomiphene citrate, with twin fetuses, was normal to the 2Nlh week. Otherwise, the patient's medical history was unremarkable. While in the hospital, a rapid weight gain occurred: protein in the urine increased from 4. S g/ da> to 9.5 g/ day. and uric acid increased to S. S mg'. r. Because of the rapid deterioration in preeclampsia, and because of a difference of 400 g in the growth of the fetuses ( 1200 g vs. SOO g). treatment with 12 mg betamethasone twice daily anil parenteral magnesium sulfate were initialed on the ninth day. That evening, the patient had a headache, went to sleep, and woke up in the morning sightless in both eves. Blood pressure was 150/ 90; physical and neurologic findings were normal. Ophthalmologic examination showed full orientation, but no light perception in either eve; pupils equal at 3 mm and briskly reactive to light, bindings in a ophthalmoscopy examination were normal. Laboratory results were normal for serum chemistrv. complete blood count, platelet count, liver functions, prothrombin time, partial thromboplastin time, and fibrinogen including anlicardiolipin antibodies. An emergency brain CT scan was performed to rule out intracranial hemorrhage. Hmcrgency cesarean delivery was performed, and the placenta was submitted for pathologic examination. Twelve hours after the operation, the patient's vision improved graduallv and linallv returned to normal. The results of a neuro- ophthalmologic exnmina- I6J 164 A. KISI. ER i: r ,\ i.. lion 3 clays after delivery were visual acuity of 20/ 20 in both eyes; Ishihara color plates. 12/ 12; visual fields, normal; visual- evoked potential, normal. An MR I was performed 4 days after delivery to delect any residual dam-aye. The (" I" scan showed low- density areas in both occipital lobes ( big. I). The results of MRI in T2- weighled images showed a high- intensity signal in the occipital areas along the gray- while junction. Some T2 signals were lateral to the basal ganglia on both sides ( big. 2). Pathologic examination of the placenta showed a di-chorionic diamniotie fused preterm twin placenta weighing 450 g. Grossly, no infarcts were noted. Histologic sections showed evidence of decidual vasculopalhy: Numerous vessels in the decidua capsularis had persistent muscular wall in the tunica media, and several vessels showed acute athcrosis- fibrinoid necrosis of the vascular wall associated with foamy macrophages. This lesion was the result of intimal and medial damage ( bigs. 3 and 4); Focally. plasma cell vasculitis was found. Also noted were plasma cell infiltration in the decidua. villitis of anchoring villi, and focal hemosiderin- laden macrophages in the membranes. These lesions indicated uteroplacental vascular pathology and chronic inflammation. DISCUSSION The cause ami pathogenesis of preeclampsia arc not fully understood. However, preeclampsia is clearly dependent on the presence of intact placental tissue and resolves after the evacuation of the uterus. The precise pathogenesis of hypertensive encephalopathy occurring in severe preeclampsia and eclampsia are as yet uncertain ( II). Because the clinical manifestations are usually reversible, vascular spasm producing transient ischemia has been suggested to explain the development of cortical blindness in patients with preeclampsia ( 8). Several theories have been put forth to explain this phenomenon, bdvinsson el al. ( 6) observed that the anterior circulation is well innervated, whereas the posterior circulation is FIG. 1. Computed tomographic brain scan, showing low densities in both parietal lobes. ./ NnimOplllhalmiil. Vol. IS. A'o. .(, I<)' JS FIG. 2. T2- weighted magnetic resonance scan, showing a high signal in the occipital areas along the gray- white junction. In addition there are a few T2 signals lateral to the basal ganglia on both sides. less so. a finding corroborated by the many lesions they detected in the occipital lobe by neuroimaging. Schwartz et al. ( 12) suggested two possible mechanisms by which normal cerebral autoregulation may be overcome: over-regulation, in which severe cerebral vasoconstriction causes lesions in the occipital region, and breakdown, in which the loss of cerebral vascular regulation with resultant dilatation causes focal vasogenic edema. Schobel et al. ( I) have theorized that the findings of an almost parallel decrease in sympathetic activity and blood pressure after delivery are highly suggestive of a neurogenic component in preeclampsia. They have suggested that augmented sympathetic vasoconstrictor activity is an important mechanism of the increase in vascular resistance FIG. 3. Decidual arteropathy in decidua capsularis attached to placental membranes. Small arteries with persistent smooth muscle in their walls, evidence of the absence of physiological change ( short arrows). A large decidual artery with acute athero-sis ( long arrow) ( hematoxylin- eosin; magnification, x100). CORTICAL BLINDNESS IN PREECLAMPSIA 165 FIG. 4. Acute atherosis in the decidual artery. Fibrinoid necrosis of the wall ( short arrow) associated with foamy ( lipid- laden) macrophages ( hematoxylin- eosin; magnification, x400; long arrows). characteristic of preeclampsia. In normal pregnancy, the endometrial spiral arterioles are invaded by trophoblast cells, replacing the smooth muscle and elastic fibers in the tunica media. This alteration results in rigid vascular channels that are unresponsive to normal vasoregulation, a physiologic change essential for adequate perfusion of the placenta. The absence of or an incomplete physiologic change in the decidual vessels causes decreased uteroplacental perfusion and development of preeclampsia. Placental ischemia stimulates secretion of vasoconstrictor substances ( thromboxane, angiotensin A, endo-thelin), and of vasodilator inhibiting agents ( prostaglandin I2, prostaglandin E2, nitric oxide). Therefore, the clinical syndrome of preeclampsia includes manifestation of disseminated intravascular coagulation and hypertension, secondary to endothelial damage ( 12- 15). We have been unable to find reports in the literature in which the association between placental lesions and the severity of the clinical picture in general or the neurologic picture in particular are examined. In the present case, we found composite evidence of vascular endothelial damage. Despite normal coagulation function, both proteinuria, suggestive of microthrombi in the glomerular capillaries, and acute atherosis in the histologic section of the placenta indicate endothelial damage and activation of the coagulation cascade. In conclusion, recent thrombosis observed in the histologic section of the placenta, ischemic changes in the brain seen in CT and MR1 scans, and severe proteinuria clinically suggest vascular endothelial damage as the underlying mechanism of this case of precclampsia- related transient cortical blindness. Acknowledgment: The authors thank Professor Kathleen B. Digre for her help in reviewing and analyzing the ncuroimages in this case. REFERENCES 1. Schobel HP, Fischer T, Hcuszer K, Geigcr H, Schmicdcr RE. Preeclampsia: a state of sympathetic overactivity. N Engl J Med 1966; 335: 1480- 5. 2. Valluri S, Adclberg DA, Curtis RS, Oik R. I. Diagnostic indocya-nine green angiography in preeclampsia. Am J Ophthalmol 1996; 122: 672- 77. 3. Digrc KB, Varncr MW, Osborn AG, Crawford S. Cranial magnetic resonance imaging in severe preeclampsia vs. eclampsia. Arch Neurol 1993 ; 50: 399- 406. 4. Salafia CM, Pczzullo JC, Lopcz- Zeno JA, Simmons S, Minior VK, Vinlzileos AM. Placental pathologic features of preterm preeclampsia. Am .1 Ohstet Gynecol 1995; 173: 1097- 1 105. 5. Jaffe G, Schatz H. Ocular manifestations of preeclampsia. Am J Ophthalmol 1987; 103: 309- 15. 6. Edvinsson L, Owman C, Sjoberg N- O. Autonomic nerves, mast cells, and amine reception in human brain vessels: histochemical and pharmacologic study. Brain lies 1976; I 15: 377- 93, 7. Vcrdile VP, Verdile LA. Post partum amaurosis. Ann Emerg Med 1991 ; 20: 3 I 1- 4. 8. Nishimura RN, Koller R. Isolated cortical blindness in pregnancy. West J Med 1982; 137: 335- 7. 9. Beeson JH, Duda EE. Computed axial tomography scan demonstration of cerebral edema in eclampsia preceded by blindness. Ohstet Gynecol 1982; 60: 529- 32. 10. Cunningham FG, Fernandez CO, Hernandez C. Blindness associated with preeclampsia and eclampsia. Am J Ohstet Gynecol 1995; 172: 1291- 8. 1 1. Sunncss JS. The pregnant woman's eye. Surv Ophthalmol 1988; 32: 219- 38. 12. Schwartz. RB, Jones MK, Kalina P, el al. Hypertensive encephalopathy: findings of CT, MR imaging, and SPECT imaging in 14 cases. Am J Roentgenol 1992; 159: 379- 83. 13. Roberts JU, Taylor RN, Musci T. I, Rogers GU, 1- Iubcl CA, McLaughlin MK. Preeclampsia: an endothelial cell disorder. Am .1 Ohstet Gynecol 1989; 161: 120( M. 14. Ferns TF. Pregnancy, preeclampsia, and the endothelial cell. N Engl J Med 1991 ; 325: 1439- 40. 15. Dekkcr GA, VanGcijn HP. Endothelial dysfunction in preeclampsia, 1: primary prevention, therapeutic perspectives../ Perinat Med 1996; 24: 99- 117. ,/ Neuro- Ophllmlmol, Vol. IS, No. .1. I99S |