| OCR Text |
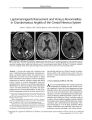
Show ORIGINAL CONTRIBUTION Linear Magnetic Resonance Enhancement and Optic Neuropathy in Primary Angiitis of the Central Nervous System Adam S. Hassan, MD, Jonathan D. Trobe, MD, Paul E. McKeever, MD, and Stephen S. Gebarski, MD Abstract: A 38- year- old woman developed incoherent mentation, tremor, ataxia, and bilateral optic disc edema with mildly depressed visual acuity, nerve fiber bundle defects, and a left afferent pupillary defect. Magnetic resonance imaging of the brain disclosed striking linear contrast enhancement radiating from the ventricular borders. Lumbar puncture showed a normal opening pressure with a lymphocytic pleocytosis and elevated protein. On the basis of these findings, the initial diagnosis was viral or post- viral meningoencephalitis and the patient was not treated. During the next 4 weeks, her condition worsened. A brain and meningeal biopsy disclosed findings typical of primary angiitis of the central nervous system. With aggressive treatment, her neurologic status and magnetic resonance imaging normalized and her optic neuropathy improved markedly. Optic neuropathy and linear magnetic resonance imaging enhancement should be recognized as features of primary angiitis of the central nervous system. ( JNeuro- Ophthalmol 2003; 23: 127- 131) Primary angiitis of the central nervous system ( PACNS) is a rare disease of unclear etiology that manifests clinically with headache, altered mentation, and a variety of focal neurologic deficits ( 1). Magnetic resonance imaging ( MRI) typically reveals multifocal white and gray matter signal abnormalities. Leptomeningeal enhancement may uncommonly occur ( 2). We present a case in which the MRI abnormalities displayed a rarely reported pattern, a radial distribution of contrast enhancement that corresponded to inflamed cerebral vessels and perivascular regions. Our patient also had bilateral optic disc edema with optic nerve Kellogg Eye Center, Departments of Ophthalmology ( ASH, JDT), Neurology ( JDT), Pathology ( Neuropathology) ( PEM), and Radiology ( Neuroradiology) ( SSG), University of Michigan Medical Center, Ann Arbor, Michigan. Address correspondence to Jonathan D. Trobe, MD, Kellogg Eye Center, 1000 Wall Street, Ann Arbor, MI 48105, USA; E- mail: jdtrobe@ umich. edu dysfunction yet normal intracranial pressure, features not previously well documented in PACNS. CASE REPORT A 38- year- old woman developed nausea, vomiting, diarrhea, myalgias, and generalized weakness in early January 2002. Within 1 week, symptoms evolved into incoherent mentation, extremity tremor, incoordination of limbs, and balance difficulty. Examination disclosed inattention, a resting tremor of the extremities, slight extremity ataxia, and mild tandem gait difficulty. Her medical history included depression, paroxysmal atrial tachycardia, and hyperlipidemia. Medications included digoxin and an oral contraceptive. Standard laboratory and toxicologic studies were negative. Brain MRI revealed diffuse abnormal contrast enhancement distributed in a linear pattern radiating from the ventricular borders into the deep white matter with surrounding T2 signal alteration. There was less striking abnormal enhancement of the leptomeninges ( Fig. 1). The subcortical white matter and gray matter were relatively spared. A lumbar puncture showed an opening pressure of 150 mm Hg with 250 white blood cells, mostly lymphocytes, a normal glucose, a slightly elevated protein at 50 mg/ dl, elevated myelin basic protein, no oligoclonal bands, and a negative cytologic study for malignancy. Polymerase chain reaction for herpes simplex and cultures of the cerebrospinal fluid, as well as serologies for rheumatologic, vasculitic, paraneoplastic, and peroxisomal disorders, were negative. The patient was given a diagnosis of viral or post-viral meningoencephalitis, and was discharged home without treatment. On examination 1 month later, the patient had greater confusion and tremor, and was complaining of decreased vision in the OS. Visual acuity was 20/ 25 OU. Pupils were equal in size, and reacted normally to direct light with an afferent defect in the OS. Ocular ductions were full with Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. J Neuro- Ophthalmol, Vol. 23, No. 2, 2003 127 JNeuro- Ophthalmol, Vol. 23, No. 2, 2003 smooth pursuit and no nystagmus. The eyes were aligned. The anterior ocular segments were normal. Ophthalmoscopy disclosed moderate bilateral optic disc edema ( Fig. 2). Formal visual fields could not be performed because of poor cooperation. She was fearful, inattentive, disoriented to time, unable to register more than two of three objects, or to remember any objects after 3 minutes. She had a shaking tremor of the head, trunk, and extremities. Deep tendon reflexes were 3+ throughout. The rest of the neurologic examination was normal. Brain MRI showed the same abnormalities noted 5 weeks earlier. A lumbar puncture had an opening pressure of 180 mm Hg and the fluid contained 150 white blood cells, a glucose of 75 mg/ dl, and a protein of 121 mg/ dl. Serologic studies identical to those performed on the earlier admission were negative. On the fifth hospital day, she became unresponsive except to deep painful stimuli, and an electroencephalogram disclosed diffuse seizure activity consistent with status epilepticus. Phenytoin treatment eliminated the seizure activity within 24 hours and restored the patient's baseline mentation. Biopsy of the right frontal cerebral cortex, underlying white matter, and overlying meninges revealed inflammation in and around vascular walls ( Fig. 3A). Immunohisto-chemical stains for CD3, CD20, CD45, and CD68 revealed Hassan et al. FIG. 1. Magnetic resonance imaging of the brain performed 2 weeks after illness onset. A: Axial T2 section through the corona radiata along the lateral ventricles. Profound signal abnormalities in the white matter are compatible with ischemic or demyelinating disease. B: Axial FLAIR at the same level. The white matter signal abnormalities are more obvious. C: Axial diffusion- weighted section at the same level. No frank restricted diffusion is seen, militating against acute or subacute infarction. D: Enhanced axial T1 section at the same level. There is striking enhancement along the perivascular regions ( arrows) and slightly along the meninges. There is no fusiform, nodular, or ring- like enhancement as would be expected with demyelinating disease. E: Enhanced coronal T1 section through the trigones of the lateral ventricles shows dramatic perivascular enhancement ( arrows). polyclonal inflammation in which T- lymphocytes predominated and B- lymphocytes and macrophages were common. Many inflammatory cells infiltrated the walls of small vessels ( Fig. 3B- D). While primarily vasocentric, a few cells occurred in brain parenchyma ( Fig. 3B, C). Intense gliosis indicated that the lesion was more than 2 weeks old ( Fig. 3E). These features substantiated the pathologic diagnosis ofPACNS. The patient was treated with intravenous methylpred-nisolone 1 gm/ d for 3 days, followed by prednisone 60 mg/ d and intravenous cyclophosphamide 125 mg/ d followed by oral cyclophosphamide 125 mg/ d. Within a month, her mentation had improved dramatically and her tremor had become less pronounced. Visual acuity was 20/ 25 OD, 20/ 30 OS, optic disc edema had lessened, and visual fields disclosed nerve fiber bundle defects binocularly, denser in the OS ( mean deviation - 3.44 dB OD, - 5.71 dB OS) ( Fig 2). During the ensuing weeks, her mentation returned to normal and her tremor disappeared. At 6 months after illness onset, visual acuity had improved to 20/ 20 OD, 20/ 25 OS, the left afferent pupil defect persisted, the optic disc edema had virtually disappeared, but visual fields still showed a defect in the OS ( Fig. 2). MRI abnormalities, both on T2 and Tl enhanced sequences, had completely resolved ( Fig 4). Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. 128 © 2003 Lippincott Williams & Wilkins OPTIC NEUROPATHY IN CNS ANGIITIS JNeuro- Ophthalmol, Vol. 23, No. 2, 2003 FIG. 2. Top: at 1.5 months after illness onset, bilateral optic disc edema is evident. Center, at 2.5 months after illness onset, optic disc edema has lessened and visual fields show defects more prominent in the OS. Bottom: at 7.5 months after illness onset, optic disc edema is gone, but a left visual field defect persists. DISCUSSION Two features of our patient's PACNS have not been well documented: the linear MRI enhancement and the optic neuropathy. Although MRI abnormalities are present in over 95% of histologically confirmed cases of PACNS ( 3,4), they are only 36% specific to PACNS ( 5). The most common findings are focal signals abnormalities with characteristics of multiple bilateral cerebral infarcts involving gray and deep white matter ( 6,7). Leptomeningeal enhancement may be associated with the parenchymal lesions ( 2) or be an isolated MRI finding ( 8). Less commonly, signal abnormalities have been reported to involve only the deep white matter ( 6,9). Our patient's MRI revealed striking linear contrast enhancement radiating from the ventricles into the deep white matter, distributed in a pattern suggestive of disease in the perivascular regions with surrounding T2 signal alterations. One published case ( 10) reported similar MRI findings, but they are less dramatic than in our case. The diffusion- weighted sequence did not suggest infarction. Instead, these MRI findings indicate severe but reversible perivascular inflammation causing blood- brain barrier disruption and injury of surrounding white matter. The radial distribution of these enhancing signal abnormalities follows the path of vessels. Likely to be specific for vasculitis, this configuration is not seen in the MRI scans of patients with multiple sclerosis or post- viral demyelination, which show ring- like, lobular, or fusiform signal abnormalities. Optic disc edema has been rarely described in PACNS. In the six reported cases, patients all had well-documented elevation of intracranial pressure ( 11- 16), as distinct from our patient, whose intracranial pressure was normal. There is one reported case of " bilateral optic atrophy" without further detail ( 20). Four cases of bilateral visual loss have been described secondary to cortical infarction ( 17- 19). Our patient likely had vasculitic inflammation Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. 129 JNeuro- Ophthalmol, Vol. 23, No. 2, 2003 Hassan et al. I H ;"- C <! » * - * ' • ' . J." • ;-_' " r * ' • • • ; - , ' *"*;'"'* "*"" a *• * * . I k . • - sSS^* v-', y- • " m • ^^ tt .<•'' v * s ^ 8 '-" - "^ Sw^ SE . . Si - ; \ ^ V w ' ^ ' y^ * p 2 @ ^ v ;': -' X^^.' f ^ Wll\ • J 0 m^ M FIG. 3. Cerebral biopsy 1.5 months after illness onset. A: Hematoxylin and eosin stain. Inflammation involves vessel walls. B: CD3 stain. Reactive T- lymphocytes mainly involve vessel, but some occur in brain tissue. C: CD20 stain. B- lymphocyte distribution resembles that of T-lymphocytes. D: Type IVcollagen stain. Inflammation within the vascular walls spreads apart their collagen matrix. E: Glial fibrillary acidic | protein stain. Reactive astrocytes show prominent cytoplasm and numerous stellate cellular processes. of the optic nerves equivalent to the process within the cerebrum. These abnormalities have not been reported in PACNS. The relative sparing of visual acuity suggests an " optic perineuritis" pattern, in which inflammation may be concentrated in the perioptic meninges. Curiously, had the optic neuropathy with disc edema been noted earlier in our patient, it might have given further support to the initial- and mistaken- diagnosis of post- viral demyelination, in which optic neuropathy is common. In reporting this case, we wish to emphasize that these MRI and optic disc findings should not be presumed to be signs of a primary demyelinating illness, in which treatment may not reverse the deficits, but should prompt a FIG. 4. Axial T1 enhanced brain MRI performed at 7.5 months after illness onset shows complete resolution. consideration of PACNS, in which aggressive treatment may restore vision and be life- saving. REFERENCES 1. Calabrese LH, Duna GF, Lie JT. Vasculitis in the central nervous system. Arthiritis Rheum 1997; 40( 7): 1189- 201. 2. Harris KG, Tran DD, Sickels WJ, et al. Diagnosing Intracranial Vasculitis: The Roles of MR and Angiography. AJNR 1994; 15: 317- 30. 3. George T, Duna GF, Rybicki L, et al: A reappraisal of primary angiitis of the central nervous system: pathologically versus angio-graphically defined cases ( abstract). Arthritis Rheum 1995; 38( suppl 9): S340. 4. Stone JH, Pomper MG, Roubenoff R, et al: Sensitivities of noninvasive tests for central nervous system vasculitis: a comparison of lumbar puncture, computed tomography, and magnetic resonance imaging J Rheumatol 1994; 21: 1277- 82. 5. Duna GF, Calabrese LH. Limitations of invasive modalities in the diagnosis of primary angiitis of the central nervous system J Rheumatol 1995; 22: 662- 7. 6. Greenan TJ, Grossman RI, Goldberg HI. Cerebral Vasculitis: MR imaging and angiographic correlation. Radiology 1992; 182: 65- 72. 7. Pierot L, Chiras J, Debussche- Depriester C, et al. Intracerebral stenosing arteriopathies: contribution of three radiological techniques to the diagnosis. JNeuroradiol 1991 ; 18: 32^ 48. 8. Negishi C, Sze G. Vasculitis presenting as primary leptomeningeal enhancement with minimal parenchymal findings. AJNR 1993; 14: 26- 8. 9. Finelli PF, Onyiuke HC, Uphoff DF. Idiopathic granulomatous angiitis of the CNS manifesting as diffuse white matter disease. Neurology 1997; 49: 1696- 9. 10. Shoemaker EI, Lin ZS, Rae- Grant AD, et al. Primary angiitis of the central nervous system: unusual MR appearance. AJNR 1994; 15: 331^ 1. 11. Castleman B, Towne VW. Case records of the Massachusetts General Hospital. Case 41152. N Engl J Med 1955; 252: 634- 41. 12. Cravioto H, Feigin I. Noninfectious granulomatous angiitis with a predilection for the nervous system. Neurology 1959; 9: 599- 609. 13. Hughes JT, Brownell B. Granulomatous Giant- celled aniitis of the central nervous system. Neurology 1966; 16: 293- 8. Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. 130 © 2003 Lippincott Williams & Wilkins OPTIC NEUROPATHY IN CNS ANGIITIS JNeuro- Ophthalmol, Vol. 23, No. 2, 2003 14. Kolodny EH, Rebeiz JJ, Caviness VS Jr, et al. Granulomatous angiitis of the central nervous system. Arch Neurol 1968; 19: 510- 24. 15. Burger PC, Burch JG, Vogel FS. Granulomatous angiitis: an unusual etiology of stroke. Stroke 1977; 8: 29- 35. 16. Rawlinson DG, Braun CW. Granulomatous angiitis of the nervous system first seen as relapsing myelopathy. Arch Neurol 1981 ; 38: 129- 31. 17. Sabharwal UK, Keogh LH, Weisman MH, et al. Granulomatous Angiitis of the nervous system: case report and review of the literature. Arthritis Rheum 1982; 25: 342- 5. 18. Case records of the Massachusetts General Hospital. Case 35- 1985. N Engl J Med 1985; 313: 566- 75. 19. Abu- Shakra M, Khraishi M, Grosman J, et al. Primary angiitis of the CNS diagnosed by angiography. QJM 1994; 87: 351- 8. 20. Jelliger K. Giant cell granulomatous angiits of the central nervous system. J Neurol 1977; 215: 175- 90. Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. 131 |