| OCR Text |
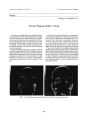
Show Journal of Neuro- Ophthalmology 17( 4): 223- 225, 1997. © 1997 Lippincott- Raven Publishers, Philadelphia Functional Magnetic Resonance Imaging of Visual Function in Postpapilledema Optic Atrophy Atsushi Miki, Takashi Nakajima, Hiruma Hasebe, and Haruki Abe Summary: We studied a girl with intraventricular brain tumor who developed postpapilledema optic atrophy and severe concentric visual field constriction in both eyes. The patient had mild symptoms despite severe visual field loss. We performed functional magnetic resonance imaging using two kinds of visual stimulation to assess her residual visual function. The functional magnetic resonance imaging demonstrated that the activity in her association visual cortex was largely intact bilaterally, although the activation of the calcarine cortex was decreased in the left calcarine cortex. Her activity in the visual cortex seemed to correspond well to her visual symptom. Functional magnetic resonance imaging may be useful in objectively documenting residual visual function in patients with severe visual loss. Key Words: Functional magnetic resonance imaging- Postpapilledema optic atrophy- Visual cortex- Visual function. Central neurocytoma is an uncommon tumor often found in young adults, and it was first described by Has-soun et al. in 1982 ( 1). The tumor can cause increased intracranial pressure because it is grown typically in the lateral ventricles ( 1). We studied a patient with papilledema caused by central neurocytoma who had severe visual field defects in both eyes. CASE REPORT A 17- year- old girl with a 10- month history of anorexia, headache, nausea, and vomiting was examined on February 27, 1995. Her visual acuity was 20/ 40 in the right eye and 20/ 200 in the left eye. Intraocular pressures were 18 mm Hg in the right eye and 17 mm Hg in the left eye. Ocular movements were normal. Visual fields snowed marked visual field contraction in both eyes. Ocular fundus showed marked papilledema with Paton's Manuscript received November 26, 1996; accepted June 20, 1997. From the Department of Ophthalmology, Niigata University School of Medicine ( A. Miki, H. Hasebe, H. Abe), and the Department of Neurology, National Saigata Hospital ( T. Nakajima), Niigata, Japan. Address correspondence and reprint requests to Dr. Atsushi Miki, Department of Ophthalmology, Niigata University School of Medicine, 1- 757 Asahimachi- dori, Niigata 951, Japan. lines. The presence of a space- occupying lesion was suspected from these signs and symptoms. Emergency magnetic resonance imaging ( MRI) scans disclosed a huge intraventricular tumor with hydrocephalus. The tumor involved both lateral ventricles, but it primarily affected the left lateral ventricle. The patient was admitted immediately and underwent a resection of the tumor by the frontal transcortical approach. A pathologic diagnosis of central neurocytoma was made. Computed tomography performed postoperatively showed a residual tumor in the lateral ventricles. The tumor could not be totally removed despite a second surgical procedure similar to the first one. After tumor removal, papilledema showed resolution gradually, but optic atrophy developed ( Fig. 1). Her visual acuity improved to 20/ 30 in both eyes, despite persistent concentric visual field contraction ( Fig. 2). She had difficulty in walking forward without aid, particularly in a dark room. However, when not moving, she had no symptoms after these operations. Functional MRI ( 2,3) was performed using a 1.5- T system ( MAGNEX 150HP, Shimadzu Corporation, Kyoto, Japan). Informed consent was obtained from the patient before the study, and all procedures were approved by the Human Investigation Committee of the National Saigata Hospital, Niigata, Japan. The patient had no symptoms or signs suggesting the increased intracranial pressure at the time of these studies. Binocular visual stimulation was accomplished using an 8- Hz flashing white light placed in front of the patient's face via glass fiber optics and an 8- Hz reversing checkerboard stimulus by means of a prism glass ( 4,5). The visual angles of the checkerboard stimulus were 13 degrees vertical and 20 degrees horizontal, with a check size of 1 degree. The stray lights illuminated the full visual fields during both visual stimuli. During the checkerboard visual stimulation periods, the patients viewed the checkerboard through a prism glass and were required to fixate at a fixation point in the center of the screen. Tl- weighted and T2*- weighted MRIs were oriented parallel to the calcarine fissure and were acquired on the same slice with a thickness of 6 mm. Functional images were obtained using a small tip angle gradient- echo sequence ( field of view = 230 mm; imaging matrix = 128 x 128 223 A. MIKIETAL. FIG. 1. Stereo fundus photographs taken on June 8, 1995, showing reducing papilledema but developing optic atrophy. A, right eye; B, left eye. pixels; repetition time = 112 ms; echo time = 60 ms; flip angle = 40 degrees; number of excitations = 1). During the resting periods no visual stimulus was presented. A series of three resting and three stimulation periods was repeated three times to generate a total of 18 images. Special care was taken to immobilize the patient's head to minimize motion artifacts. Data were analyzed by Student's paired t test on a pixel- by- pixel basis, and significantly activated regions, defined as the pixels with a p value of < 0.01, were superimposed on the Tl-weighted image. During the periods of flash stimulation, the activated area was small and almost no activation was observed in the left primary visual cortex, corresponding to the concentric visual field constriction and the dense defects of right visual field ( Fig. 3, A). However, the activated area extended to the lateral occipital cortex when the patient watched the alternating checkerboard pattern ( Fig. 3, B). DISCUSSION Chronic papilledema produces visual field constriction, and its visual prognosis is often poor. The patient described above had developed optic atrophy despite removal of the tumor. Irreversible changes seemed to have occurred in the optic nerve because she had already had marked visual field loss with presumably prolonged papilledema at the first consultation in our hospital. However, despite the severe visual loss, no symptom referable to higher visual function was observed. Because the surgeries were performed using the frontal transcortical approach, it did not seem that the posterior afferent visual system had been damaged during these procedures. Functional MRI is one of the recent functional imaging techniques of the brain. It can precisely localize regional brain activity with fine spatial resolution. Recent functional MRI has shown selective activation of visual cortex during visual stimulation ( 2- 9). Whereas the white flashing stimulus has been demonstrated to produce the activation in the primary visual cortex mainly with limited involvement of the extrastri-ate cortex ( 10), the reversing checkerboard is shown to produce activation in and outside the primary visual cortex ( 8- 10). The patient had decreased activity in the portion of the primary visual cortex corresponding to the visual field defects but had relatively preserved activity in the portion of lateral occipital cortex corresponding to her visual symptom. The patient's difficulty in walking seems to occur in the setting of severe visual field contraction. Because visual loss in the patient's right hemi-field was dense, the activation of the left association visual cortex implied that the fibers of the corpus callo-sum, linking the right and left visual cortex, was preserved at least in part. To our knowledge, there has been no report concerning functional MRI in patients with concentric visual field constriction. This case suggests that the higher visual function may be preserved until the end stage of the disease with concentric visual field constriction. FIG. 2. The threshold perimetry ( Humphrey 30- 2) showed constricted visual fields. The defects were denser on the right visual field in both eyes. Both eyes had no fixation losses, and no false- positive and false-negative errors. Visual field defects were reproduced with the Humphrey 10- 2 program and with the Gold-mann perimetry. Left, left eye; right, right eye. J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 POSTPAPILLEDEMA OPTIC ATROPHY 225 FIG. 3. Functional magnetic resonance images during the flash stimulation ( A) and during the checkerboard stimulation ( B). The patient's right is on the left. Pixels with signal increases of p < 0.01 were superimposed on the anatomic images. Correlating well with the patient's symptom, cerebral activity in the prestriate cortex was preserved during the checkerboard stimulation. REFERENCES 1. Hassoun J, Soylemezoglu F, Gambarelli D, Figarella- Branger D, von Amnion K, Kleihues P. Central neurocytoma: a synopsis of clinical and histological features. Brain Pathol 1993; 3: 297- 306. 2. Ogawa S, Tank DW, Menon R, et al. Intrinsic signal changes accompanying sensory stimulation: functional brain mapping with magnetic resonance imaging. Proc Natl Acad Sci USA 1992; 89: 5951- 5. 3. Kwong KK, Belliveau JW, Chesler DA, et al. Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc Natl Acad Sci USA 1992; 89: 5675- 9. 4. Miki A, Nakajima T, Fujita M, Takagi M, Abe H. Functional magnetic resonance imaging in homonymous hemianopsia. Am J Ophthalmol 1996; 121: 258- 66. 5. Miki A, Nakajima T, Takagi M, Shirakashi M, Abe H. Detection of visual dysfunction in optic atrophy by functional magnetic reso- 10. nance imaging during monocular visual stimulation. Am J Ophthalmol 1996; 122: 404- 15. Schneider W, Noll DC, Cohen JD. Functional topographic mapping of the cortical ribbon in human vision with conventional MRI scanners. Nature 1993; 365: 150- 3. Tootell RBH, Reppas JB, Kwong KK, et al. Functional analysis of human MT and related visual cortical areas using magnetic resonance imaging. J Neurosci 1995; 15: 3215- 30. Sereno MI, Dale AM, Reppas JB, et al. Borders of multiple visual areas in humans revealed by functional magnetic resonance imaging. Science 1995; 268: 889- 93. DeYoe EA, Carman GJ, Bandettini P, et al. Mapping striate and extrastriate visual areas in human cerebral cortex. Proc Natl Acad Sci USA 1996; 93: 2382- 6. Phelps ME, Mazziotta JC, Kuhl DE, et al. Tomographic mapping of human cerebral metabolism: visual stimulation and deprivation. Neurology 1981; 31: 517- 29. J Neuro- Ophthalmol, Vol. 17, No. 4, 1997 |